Clinical research careers in India include roles such as Clinical Research Associate, Clinical Data Coordinator, Pharmacovigilance Associate, and Clinical Trial Assistant. Growing clinical trials, AI adoption, and global outsourcing are creating increasing opportunities across CROs, hospitals, pharmaceutical companies, and research organizations.
The clinical research industry is entering a new phase of growth. As pharmaceutical companies accelerate drug development, biotechnology innovation expands, and global clinical trials become more complex, the demand for skilled clinical research professionals continues to rise.
The industry today offers far more than traditional clinical trial roles. Opportunities now exist across clinical operations, pharmacovigilance, clinical data management, regulatory affairs, medical writing, and emerging technology-driven functions that support modern healthcare research.
For life science, pharmacy, nursing, and healthcare graduates, clinical research has become one of the most promising career pathways, offering diverse career opportunities in clinical research, strong career progression potential, and exposure to the global healthcare ecosystem. The growing number of clinical research jobs in India reflects the industry’s increasing need for qualified professionals across multiple domains.
In this article, we explore the top clinical research careers in 2026, their responsibilities, growth potential, and why they continue to attract professionals from across the healthcare and life sciences sectors.
Clinical Research Industry Growth in India
The clinical research industry is becoming increasingly important to the future of healthcare, supporting the development of new drugs, therapies, and medical innovations. As research activities expand worldwide, the global clinical trials market is projected to grow from USD 89 billion in 2025 to USD 158.4 billion by 2033, reflecting the rising investment in clinical development and patient-centered research.
India is playing a growing role in this transformation. With its strong pharmaceutical sector, expanding biotechnology ecosystem, and large pool of research talent, the country’s clinical trials market was valued at USD 1.42 billion in 2024 and is expected to grow steadily through 2030 at a CAGR of 8.0%.
This momentum is creating new opportunities across clinical operations, pharmacovigilance, clinical data management, regulatory affairs, and other specialized areas. As pharmaceutical companies, biotechnology firms, and CROs continue to strengthen their research capabilities, the demand for skilled clinical research professionals is expected to remain on an upward trajectory.
Factors Affecting Clinical Research Careers
The demand for clinical research careers is growing due to industry expansion, technology adoption, and global outsourcing. Today, choosing the best career in clinical research depends on practical skills, regulatory knowledge, and a strong understanding of clinical trial processes. As organizations continue to expand research activities worldwide, opportunities for specialized clinical trial jobs are increasing across multiple domains within the industry.
- Technological Advancements
AI, machine learning, and data analytics are transforming clinical trials through faster patient recruitment, monitoring, and data analysis, creating new opportunities in clinical trial jobs and healthcare research.
- Regulatory Changes
Global guidelines like ICH-GCP and FDA regulations are increasing demand for professionals skilled in compliance, data integrity, and audit readiness.
- Industry Growth
Pharmaceutical, biotechnology, hospital, and medical device industries are growing at nearly 5–6% annually, increasing demand for clinical trials and expanding clinical research career opportunities.
- Outsourcing Growth
India has become a major hub for CROs, clinical data management, and pharmacovigilance services, driving large-scale hiring across clinical trial jobs.
- Importance of Soft Skills
Communication, teamwork, and adaptability are highly valued in globally connected and remote clinical research environments.
Clinical Research
Build practical skills required for alternative career paths in the pharmaceutical and healthcare industries. This program introduces clinical trial processes, regulatory documentation, drug safety monitoring, and research data management used in global clinical research operations.
Most In-Demand Clinical Research Careers in 2026
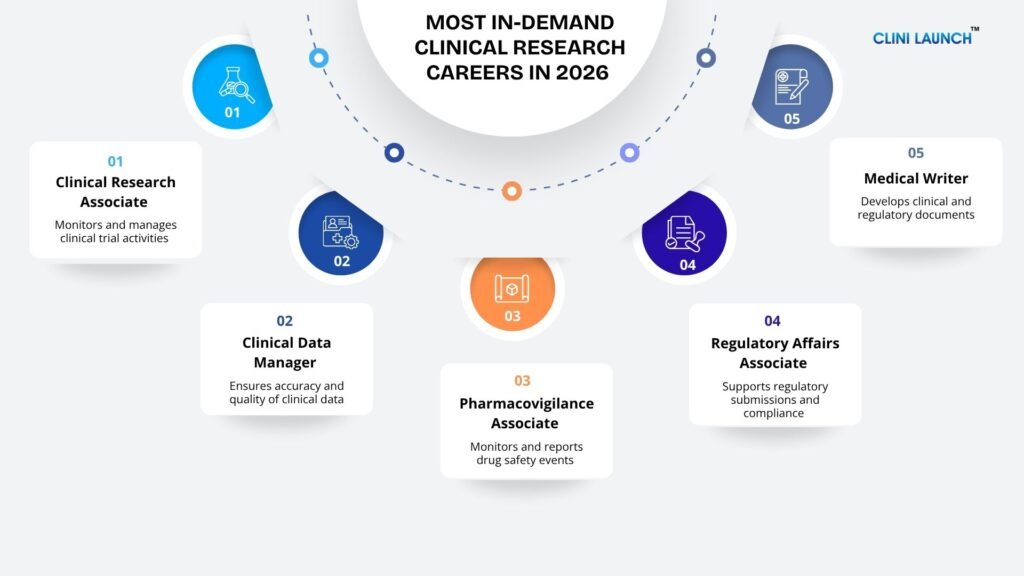
- Clinical Research Associate
- Clinical Data Management
- Pharmacovigilance Associate
- Regulatory Affairs Associate
- Medical writer
- Biostatistician
- Clinical Trial Manager
- Clinical Research Coordinator
- Clinical Quality Associate
- Clinical Operations Assistant
1. Clinical Research Associate
A Clinical Research Associate is responsible for monitoring and managing clinical trials to ensure they are conducted safely, ethically, and according to approved study protocols and regulatory guidelines. CRAs review patient data, verify clinical trial documentation, coordinate with hospitals and research sites, ensure compliance with standards such as ICH-GCP and FDA regulations, and help maintain the accuracy and quality of clinical trial data throughout the study.
CRA is considered one of the most promising career paths in the clinical research industry, offering opportunities to work with pharmaceutical companies, biotech firms, CROs, and global clinical trials, with strong career growth and increasing industry demand.
Responsibilities
- Monitoring clinical trials
- Ensuring protocol compliance
- Reviewing patient data
- Maintaining trial documentation
2. Clinical Data Management
A Clinical Data Management is responsible for managing and maintaining clinical trial data to ensure it is accurate, complete, and compliant with approved study protocols and regulatory guidelines. Clinical Data Associate reviews clinical datasets, perform data validation, identify inconsistencies, maintain electronic clinical databases, and support the quality and reliability of clinical trial data throughout the research process.
Clinical Data Management is a vital function in modern clinical research, ensuring that clinical trial data is accurate, complete, and ready for analysis and regulatory review. As clinical studies become more technology-driven, pharmaceutical companies, biotech firms, and CROs continue to seek skilled data management professionals, making it a promising career path with strong growth opportunities.
Responsibilities
- Managing clinical trial data
- Data validation and data cleaning
- Reviewing clinical datasets
- Maintaining electronic clinical databases
- Identifying data inconsistencies
- Ensuring data quality and regulatory compliance
3. Pharmacovigilance Associate
A Pharmacovigilance Associate, also known as a Drug Safety Associate, is responsible for monitoring the safety of medicines during clinical trials and after drug approval. PV professionals collect, review, document, and report adverse drug reactions (ADRs) to ensure patient safety and regulatory compliance. The role involves maintaining safety databases, reviewing case reports, supporting regulatory submissions, and ensuring compliance with global pharmacovigilance guidelines. For freshers, Pharmacovigilance offers structured exposure to drug safety operations and pharmaceutical workflows. It is considered a strong and stable career path in the pharmaceutical industry, with opportunities to grow into roles such as Senior PV Associate, Drug Safety Specialist, PV Scientist, Medical Reviewer, and Drug Safety Manager globally.
Responsibilities
- Monitoring adverse drug reactions (ADRs)
- Drug safety reporting
- Reviewing and documenting safety cases
- Maintaining pharmacovigilance databases
- Regulatory compliance and safety monitoring
4. Regulatory Affairs Associate
A Regulatory Affairs Associate is responsible for ensuring that pharmaceutical, biotech, and healthcare products comply with national and international regulatory guidelines. Regulatory professionals prepare and maintain regulatory documents, support product submissions, coordinate with regulatory authorities, and help organizations meet healthcare compliance standards throughout the product lifecycle.
The role involves managing regulatory documentation, tracking regulatory updates, and supporting approvals for drugs, medical devices, and clinical research activities. For freshers, Regulatory Affairs offers structured exposure to compliance processes and global healthcare regulations. It is considered a stable and growing career path in the pharmaceutical industry, with opportunities to progress into roles such as Regulatory Specialist, Regulatory Manager, and Global Regulatory Affairs professional across pharmaceutical, biotech, and CRO sectors.
Responsibilities:
- Regulatory documentation
- Product submissions and approvals
- Compliance management
- Regulatory reporting
- Coordinating with regulatory authorities
- Tracking healthcare regulations and updates
5. Medical writer
A Medical Writer (Clinical & Regulatory) is responsible for creating clear, accurate, and scientifically structured documents used in clinical trials and regulatory submissions. Medical Writers prepare documents such as clinical study reports (CSRs), protocols, investigator brochures, patient information materials, and regulatory dossiers while ensuring compliance with ICH-GCP and FDA guidelines.
The role involves interpreting clinical and research data, collaborating with clinical, regulatory, and pharmacovigilance teams, and maintaining consistency across scientific documents. For freshers, Medical Writing offers a stable and growing career path that combines healthcare knowledge with communication skills, without requiring patient-facing work or site travel. Career progression can lead to roles such as Senior Medical Writer, Regulatory Writer, , and Medical Communications Specialist.
Responsibilities
- Preparing clinical and regulatory documents
- Writing clinical study reports
- Developing protocols and investigator brochures
- Creating patient information materials
- Reviewing and interpreting clinical data
- Ensuring regulatory and scientific accuracy
6. Biostatistician
A Biostatistician or Statistical Programmer is responsible for analyzing clinical trial and healthcare data to determine the safety and effectiveness of treatments. These professionals apply statistical methods, prepare analysis-ready datasets, generate reports, and support evidence-based decisions used in clinical research and regulatory submissions.
The role involves working with clinical data, performing statistical analysis, supporting trial reports, and ensuring data accuracy and compliance with global standards. For freshers, this career offers strong exposure to healthcare analytics, clinical trial design, and statistical software while building practical analytical skills. It is considered a high-demand and stable career path in pharmaceutical companies, CROs, biotech firms, and healthcare analytics, with growth opportunities into senior biostatistics, statistical programming, and clinical data science roles.
Responsibilities
- Clinical trial data analysis
- Statistical programming
- Data validation and interpretation
- Regulatory reporting
- Working with statistical software
7. Clinical Trial Manager
A Clinical Trial Manager oversees the planning, execution, and successful completion of clinical trials while ensuring compliance with study protocols, regulatory requirements, timelines, and budgets. The role involves coordinating cross-functional teams, managing study sites, monitoring trial progress, resolving operational challenges, and ensuring high-quality trial delivery. Clinical Trial Managers work closely with sponsors, CROs, investigators, CRAs, and regulatory teams to keep studies on track and audit-ready. This is a leadership role within clinical operations and offers opportunities to progress into positions such as Senior Clinical Trial Manager, Clinical Project Manager, Clinical Operations Manager, and Director of Clinical Operations across pharmaceutical, biotechnology, and contract research
Responsibilities
- Clinical trial management
- Study planning and execution
- Site and vendor management
- Regulatory compliance
- Budget and timeline management
8. Clinical Research Coordinator
A Clinical Research Coordinator (CRC) is responsible for coordinating and supporting the day-to-day activities of clinical trials at hospitals, research centers, and healthcare institutions. They work closely with investigators, sponsors, patients, and Clinical Research Associates to ensure studies are conducted according to approved protocols, ethical guidelines, and regulatory requirements. Key responsibilities include participant recruitment, informed consent management, study documentation, scheduling patient visits, data collection, and maintaining trial records.
Clinical Research Coordinator is a valuable entry point into the clinical research industry and offers opportunities to work on diverse clinical studies. With experience, CRCs can advance into roles such as Clinical Research Associate (CRA), Clinical Trial Manager, Clinical Operations Specialist, or other leadership positions within pharmaceutical companies, CROs, and research organizations.
Responsibilities
- Coordinate day-to-day clinical trial activities.
- Recruit and manage study participants.
- Maintain study documentation and records.
- Ensure protocol and regulatory compliance.
9. Clinical Quality Associate
A Clinical Quality Associate plays a key role in ensuring that clinical research processes, documentation, and systems meet regulatory and quality standards. The role involves supporting quality assurance activities through record reviews, audit preparation, inspections, compliance monitoring, and documentation management. Clinical Quality Associates help ensure adherence to guidelines such as ICH-GCP and other regulatory requirements while working closely with clinical and quality teams to identify process improvements and maintain operational excellence. This position offers valuable exposure to quality management systems, regulatory compliance, and audit processes within the pharmaceutical, biotechnology, and clinical research sectors. Career progression can lead to opportunities in Clinical Quality Assurance, GCP Auditing, Regulatory Compliance, Quality Management, and leadership roles within quality and clinical operations.
Responsibilities
- Quality assurance and compliance
- Clinical documentation review
- GCP and regulatory compliance
- Audits and inspections
10. Clinical Operations Assistant
A Clinical Operations Assistant supports the planning, coordination, and execution of clinical trials by ensuring that study activities are conducted efficiently and in compliance with regulatory requirements. The role involves managing trial documentation, tracking study timelines, coordinating with clinical teams, monitoring trial progress, and supporting operational activities across different phases of a study. Clinical Operations Assistants help ensure adherence to protocols, ICH-GCP guidelines, and CDSCO regulations while facilitating smooth communication between sponsors, investigators, sites, and study teams.
Responsibilities
- Clinical Trial Coordination
- Study Documentation Management
- Regulatory Compliance
- Clinical Project Tracking
Comparison Table: Responsibilities, Skills, Eligibility & Salary
Compare the key responsibilities, required skills, eligibility criteria, and average salary ranges across popular career paths in Clinical Research. This overview can help you understand the opportunities available and identify the role that best matches your interests and career goals.
| Job Role | What They Do (Description) | Key Skills Required | Educational Background | Entry Level Opportunity | Average Salary in India* |
|---|---|---|---|---|---|
| Clinical Research Associate (CRA) | Monitor clinical trials and ensure protocol compliance | Monitoring, GCP, Site Management, Documentation | Life Sciences, Pharmacy, Nursing, Medical Background | Yes | ₹3–6 LPA |
| Clinical Data Management Professional | Manage, validate, and maintain clinical trial data | Data Validation, EDC Systems, Attention to Detail, SQL (optional) | Life Sciences, Pharmacy, Biotechnology | Yes | ₹3–5.5 LPA |
| Pharmacovigilance Associate | Monitor and report drug safety information | Case Processing, Drug Safety, Regulatory Guidelines | Pharmacy, Life Sciences, Medical Graduates | Yes | ₹3–5.5 LPA |
| Regulatory Affairs Associate | Prepare and manage regulatory submissions | Regulatory Documentation, Compliance, Communication | Pharmacy, Life Sciences | Yes | ₹3.5–6 LPA |
| Medical Writer | Create scientific and regulatory documents | Scientific Writing, Literature Review, Communication | Life Sciences, Pharmacy, Medical Background | Yes | ₹3–6 LPA |
| Biostatistician | Analyze clinical trial data using statistical methods | Statistics, SAS, R, Data Analysis | Statistics, Mathematics, Biostatistics, Life Sciences | Limited Freshers | ₹4–8 LPA |
| Clinical Trial Manager | Oversee planning and execution of clinical trials | Project Management, Leadership, Budgeting, Trial Operations | Life Sciences, Pharmacy + Experience | No (Experienced Role) | ₹12–25+ LPA |
| Clinical Research Coordinator (CRC) | Coordinate patient visits and trial activities at study sites | Patient Coordination, Documentation, Communication, GCP | Life Sciences, Pharmacy, Nursing | Yes | ₹2.5–5 LPA |
| Clinical Quality Associate | Ensure quality and compliance in clinical research processes | Auditing, Quality Systems, CAPA, Compliance | Life Sciences, Pharmacy | Yes | ₹3–6 LPA |
Ready to start your clinical research career? Explore industry-focused training, gain practical skills, and prepare for opportunities in clinical trials, pharmacovigilance, clinical data management, and regulatory affairs.
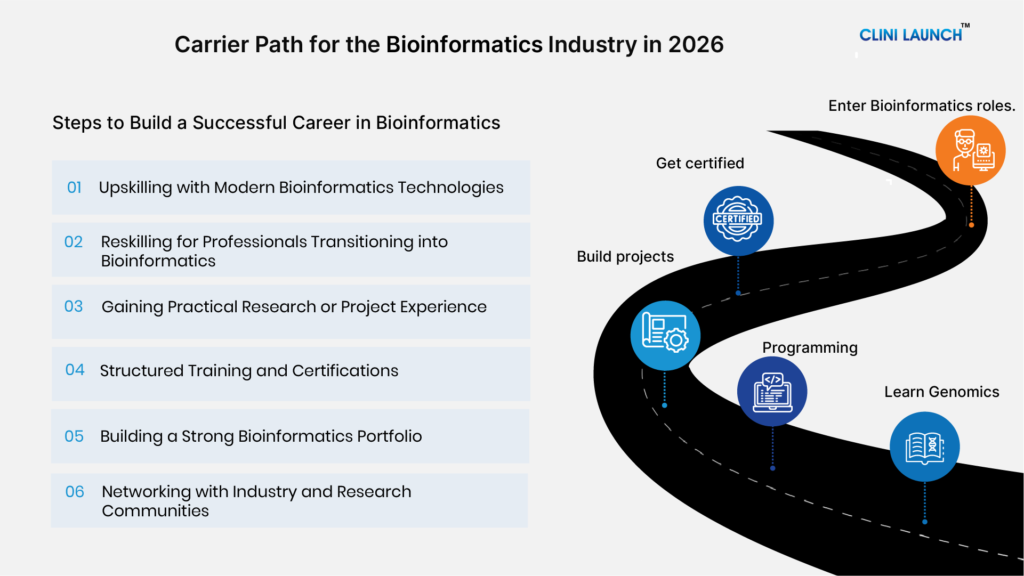
How to Start a Clinical Research Career
- Clinical research plays a critical role in bringing new therapies and healthcare innovations to patients while ensuring safety and regulatory compliance.
- The industry offers opportunities across domains such as Clinical Operations, Pharmacovigilance, Clinical Data Management, and Regulatory Affairs.
- Building a successful clinical research career requires a combination of industry-relevant knowledge, practical training, and job-ready skills.