Why Bioinformatics Career Opportunities are Growing in 2026
Bioinformatics is emerging as one of the most transformative and fast-growing careers at the intersection of biology, data science, and computing, particularly as large-scale genomic research initiatives continue to expand worldwide. Fueled by advancements in genomics, proteomics, machine learning, and precision medicine, the bioinformatics industry is experiencing robust global expansion. According to industry research, the global bioinformatics market is projected to grow from USD 17.66 billion in 2025 to USD 25.87 billion by 2030, reflecting sustained growth as more companies adopt advanced biological data analysis tools and technologies. Many learners are also exploring bioinformatics online to build flexible, industry-relevant skills.
Advances in genomics, AI-enabled biological analysis, and precision medicine are rapidly expanding career opportunities in bioinformatics across pharmaceuticals, biotechnology, healthcare, and research.
This blog explores the top bioinformatics careers for 2026 across beginners, intermediate, and advanced levels to help life science and technology graduates identify practical, future-ready career paths. For those interested in developing practical skills in this field, structured programs such as an advanced bioinformatics course like the Advanced Diploma in Bioinformatics provide hands-on exposure to real-world biological data analysis. This makes it increasingly important for building practical, job-ready skills.
Top Factors Driving Bioinformatics Career Growth in 2026
Bioinformatics careers are growing rapidly because companies and research institutes are handling vast amounts of biological data from genomics, proteomics, and health studies, and they need specialists to interpret it. The global bioinformatics market is projected to expand significantly in the coming years, with estimates showing it could grow at double-digit rates through 2030 as demand increases for data-driven discoveries. This growth reflects the central role bioinformatics plays in modern biological sciences and healthcare.
For beginners, this shift has created more structured entry and mid-level roles within bioinformatics teams. As organizations build layered teams, early-career professionals support data processing and analysis under defined workflows, while senior experts focus on strategy and translation. This structure makes bioinformatics a practical and sustainable career path for those interested in combining biology with data and technology. This growth is also increasing the demand for structured bioinformatics training programs.
Why are bioinformatics professionals in high demand globally?
The demand for bioinformatics professionals has grown rapidly as healthcare, biotechnology, and life sciences industries become increasingly data driven.
Bioinformatics professionals are in high demand globally because modern life sciences and healthcare are increasingly driven by large, complex biological datasets from genomics, drug discovery, clinical trials, and precision medicine. Organizations across pharmaceuticals, biotechnology, healthcare, and research rely on bioinformatics expertise to analyze, validate, and interpret this data, while professionals who combine biological knowledge with computational skills remain limited, creating a persistent global demand for bioinformatics talent.
Where do Bioinformatics professionals work?
Bioinformatics professionals work in organizations where biological data is generated, where biological data is generated and bioinformatics solutions are applied, including:
- Pharmaceutical companies – supporting drug discovery, target identification, biomarker analysis, and data-driven R&D decisions
- Biotechnology firms – analyzing genomic, transcriptomic, and proteomic data for research and product development
- Clinical Research Organizations (CROs) – working on clinical trial data, genomics studies, and research analytics, often collaborating with professionals trained in areas such as clinical research and bioinformatics.
- Genomics and diagnostic laboratories – processing sequencing data and supporting clinical genomics reporting
- Hospitals and precision medicine centers – assisting with patient-specific genomic analysis and decision support
- Healthcare and life-science technology companies – developing platforms, pipelines, and analytics tools for biological data
- Academic and government research institutions – conducting large-scale biological, genomic, and public health research.
Skills Required for a Successful Bioinformatics Career
Bioinformatics combines biology, data science, and computational methods to solve complex problems in healthcare, biotechnology, genomics, and life sciences research. To build strong Bioinformatics career opportunities, professionals need a combination of biological understanding, programming skills, and analytical thinking.

Biology Knowledge
A strong foundation in biology is essential for anyone entering bioinformatics. Professionals need to understand concepts such as molecular biology, genetics, cell biology, microbiology, and biochemistry to interpret biological data correctly. This knowledge helps bioinformatics professionals work with DNA, RNA, proteins, disease pathways, drug discovery datasets, and clinical research data more effectively.
Python and R Programming
Programming is one of the most important technical skills in bioinformatics. Python is widely used for data processing, automation, machine learning, and handling biological datasets, while R is commonly used for statistical analysis, visualization, and genomics research. These programming languages help professionals analyze sequencing data, build bioinformatics pipelines, create visual reports, and work with large-scale biological datasets efficiently.
Genomics Knowledge
Genomics is a core area within bioinformatics and focuses on studying genes, DNA sequences, mutations, and genetic variations. Bioinformatics professionals frequently work with next-generation sequencing (NGS), genome mapping, transcriptomics, and precision medicine datasets. Understanding genomics helps professionals contribute to areas such as cancer research, personalized medicine, rare disease analysis, and biotechnology innovation.
Data Analysis Skills
Bioinformatics generates massive amounts of biological and healthcare data that must be cleaned, processed, analyzed, and interpreted accurately. Strong data analysis skills help professionals identify patterns, validate research findings, and generate meaningful biological insights. Professionals often work with datasets from clinical trials, sequencing technologies, laboratory experiments, and healthcare systems, making analytical thinking a critical skill in bioinformatics careers.
Machine Learning Basics
Machine learning is becoming increasingly important in modern bioinformatics applications. Basic knowledge of machine learning helps professionals work on predictive modeling, disease classification, drug discovery, protein structure prediction, and AI-driven healthcare research. As AI adoption grows across healthcare and biotechnology industries, machine learning skills can significantly improve career opportunities in advanced bioinformatics roles.
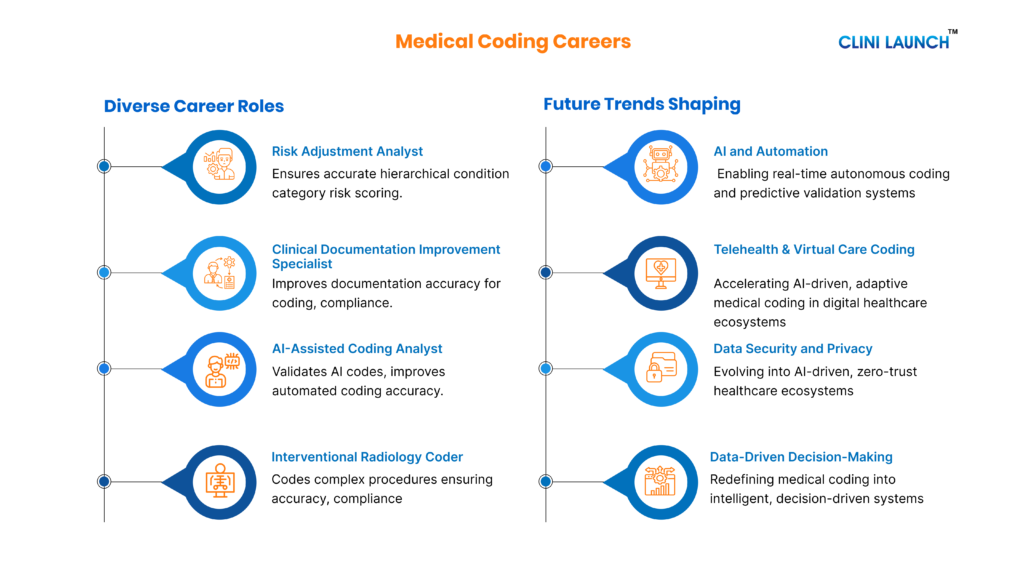
Top Bioinformatics Career Roles for 2026
Bioinformatics offers diverse career roles, from beginner-level data analysis positions to advanced scientific and leadership roles. These careers provide clear growth paths across healthcare, biotechnology, and research.
Career Path in Bioinformatics:
Bioinformatics careers typically begin with data-focused roles and progress toward research, strategy, and leadership positions as professionals gain technical and analytical experience.
A typical career path may include:
Entry-Level Roles- Bioinformatics Technician, Bioinformatics Analyst, Genomics Data Analyst, NGS Data Analyst
Mid-Level Roles- Computational Biologist, Genomics Data Scientist, Proteomics Data Analyst
Senior & Leadership Roles- Translational Bioinformatics Scientist, Principal Bioinformatics Scientist, Clinical Bioinformatics Director
With experience and specialization, professionals may also move into areas such as precision medicine, drug discovery analytics, and bioinformatics platform development.
- Bioinformatics Technician
About the Role: A Bioinformatics Technician supports genomics and bioinformatics teams by managing biological datasets and executing predefined analysis of workflows. The role focuses on ensuring that data pipelines run smoothly and that datasets are properly organized for analysis. It is a highly practical, entry-level role designed for beginners who want hands-on exposure to bioinformatics systems and workflows without deep analytical or research responsibilities.
Primary Responsibilities:
- Assist in running standard bioinformatics and sequencing workflows
- Manage and organize biological data files and datasets
- Perform basic data checks and ensure data completeness
- Support analysts and scientists during data processing tasks
Role Overview
| Category | Details |
|---|---|
| Average Salary (India) | ₹3 – ₹6 LPA (entry-level role, varies by organization and domain) |
| What They Do / Nature of Work | Support bioinformatics workflows by managing data, executing predefined processes, and assisting analysts and scientists. |
| Senior / Global Level Salary | ₹8 – ₹15+ LPA in India; USD 60,000 – 90,000 annually in global markets. |
| Growth Outlook (Next 5 Years) | Steady growth as genomics and data-driven research expand across healthcare and life sciences. |
| Job Mobility | Moderate to high opportunities across genomics labs, biotech firms, CROs, healthcare research units, and bioinformatics service providers. |
| Key Benefits | Beginner-friendly role, hands-on exposure, low entry barrier, and a strong foundation for moving into analyst or specialist roles. |
2. Bioinformatics Analyst – Career Role in 2026
About the Role: A Bioinformatics Analyst works with biological and healthcare data to identify patterns, generate insights, and support research and clinical decision-making. The role involves analyzing genomic datasets, interpreting sequencing results, and using bioinformatics tools to assist research teams in biotechnology, pharmaceuticals, healthcare, and genomics organizations. In 2026, this role continues to grow rapidly due to increasing demand for precision medicine, AI-driven healthcare, and genomic research.
Primary Responsibilities:
- Analyze genomic, proteomic, and biological datasets using bioinformatics tools
- Interpret sequencing and molecular biology data for research projects
- Support clinical and pharmaceutical research through data-driven insights
- Perform data visualization, reporting, and quality analysis
- Collaborate with researchers, scientists, and healthcare teams on bioinformatics workflows
- Maintain accuracy and integrity of biological databases and analytical results
- Assist in developing and optimizing bioinformatics pipelines and analysis methods
Role Overview
| Category | Details |
|---|---|
| Average Salary (India) | ₹4 – ₹8 LPA (entry-level to early-career professionals, depending on skills and organization) |
| What They Do / Nature of Work | Analyze biological datasets, execute validated bioinformatics pipelines, interpret results, and support research or clinical decision-making. |
| Senior / Global Level Salary | ₹15 – ₹30+ LPA in India; USD 90,000 – 140,000 annually in global markets. |
| Growth Outlook (Next 5 Years) | Strong and sustained growth driven by genomics expansion, AI-enabled research, and data-driven drug discovery. |
| Job Mobility | High opportunities across pharma, biotech, CROs, genomics labs, healthcare technology firms, and research institutions. |
| Key Benefits | Beginner-friendly entry role, strong learning curve, global applicability, and clear progression into advanced bioinformatics and genomics roles. |
- Genomics Data Analyst (Junior Level)-
About the Role: A Genomics Data Analyst (Junior Level) works with DNA-based datasets to support genomics research, diagnostics, or clinical studies. The role focuses on analyzing genomic sequencing data, identifying genetic variants, and assisting in interpretation under established workflows. It is a beginner-friendly role for individuals interested in understanding how genetic data is analyzed and applied in research and healthcare settings.
Primary Responsibilities:
- Analyze genomic sequencing data using standard analysis pipelines
- Perform quality checks on genomic datasets
- Assist in variant identification and annotation
- Support genomics research or clinical teams with analysis summaries
Role Overview
| Category | Details |
|---|---|
| Average Salary (India) | ₹4 – ₹7 LPA (entry-level to early-career professionals, depending on organization and skill set) |
| What They Do / Nature of Work | Analyze genomic data, assist in variant annotation, and support research or clinical genomics workflows. |
| Senior / Global Level Salary | ₹12 – ₹25+ LPA in India; USD 80,000 – 120,000 annually in global markets. |
| Growth Outlook (Next 5 Years) | Strong growth driven by expansion of genomics research, diagnostics, and precision medicine. |
| Job Mobility | High opportunities across genomics labs, biotech companies, diagnostic firms, CROs, and healthcare research centers. |
| Key Benefits | Entry-friendly genomics role, strong biological focus, and clear progression into advanced genomics or clinical bioinformatics careers. |
- NGS Data Analyst (Junior Level)-
About the Role: An NGS Data Analyst (Junior Level) focuses on processing and managing data generated from next-generation sequencing platforms. The role is centered on executing sequencing analysis pipelines, monitoring data quality, and ensuring that raw sequencing data is correctly processed for downstream analysis. It is a structured, workflow-driven role that serves as an ideal entry point for beginners who prefer hands-on technical work with sequencing data.
Primary Responsibilities:
- Run standard NGS data processing pipelines
- Perform quality control checks on sequencing data
- Process raw sequencing files into analysis-ready formats
- Support genomics and bioinformatics teams with clean datasets.
Role Overview
| Category | Details |
|---|---|
| Average Salary (India) | ₹4 – ₹7 LPA (entry-level to early-career professionals, varies by organization and domain) |
| What They Do / Nature of Work | Process and manage sequencing data, execute NGS pipelines, and ensure data quality for downstream analysis. |
| Senior / Global Level Salary | ₹12 – ₹25+ LPA in India; USD 80,000 – 120,000 annually in global markets. |
| Growth Outlook (Next 5 Years) | Strong growth due to widespread adoption of NGS in research, diagnostics, and clinical genomics. |
| Job Mobility | High opportunities across genomics labs, diagnostic centers, biotech firms, CROs, and sequencing service providers. |
| Key Benefits | Highly structured entry role, strong technical foundation, hands-on exposure to sequencing workflows, and a clear path into genomics or bioinformatics analyst roles. |
- Computational Biologist (Applied Level)-
About the Role: A Computational Biologist (Applied Level) works on solving biological problems using computational and statistical methods. Unlike entry-level analyst roles, this position involves designing analysis approaches, applying existing models, and independently interpreting complex biological datasets. The role bridges biology, data science, and applied research, and is typically pursued after gaining hands-on experience in bioinformatics or genomics roles. These roles contribute directly to developing scalable bioinformatics solutions for research and drug discovery.
Primary Responsibilities:
- Apply computational methods to analyze complex biological datasets
- Design and adapt analysis workflows for research or discovery projects
- Perform statistical and model-based analysis of biological data
- Interpret results in biological and experimental contexts
Role Overview
| Category | Details |
|---|---|
| Average Salary (India) | ₹8 – ₹14 LPA (mid-level professionals, depending on experience and organization) |
| What They Do / Nature of Work | Apply computational and statistical methods to analyze biological data and support research or discovery decisions. |
| Senior / Global Level Salary | ₹20 – ₹40+ LPA in India; USD 100,000 – 150,000 annually in global markets. |
| Growth Outlook (Next 5 Years) | Strong growth driven by increased use of computational biology in drug discovery, genomics, and systems biology. |
| Job Mobility | High opportunities across pharma R&D, biotech firms, genomics companies, research institutions, and health-tech organizations. |
| Key Benefits | Higher analytical ownership, intellectually challenging work, strong global demand, and a pathway to senior scientific and leadership roles. |
- Genomics Data Scientist-
About the Role A Genomics Data Scientist works on extracting insights from large-scale genomic datasets using statistical, computational, and data science methods. This role goes beyond running standard pipelines and involves designing analytical approaches, integrating genomics data with other datasets, and supporting research, clinical, or product decisions. It is an intermediate-level role typically taken after experience in genomics or bioinformatics analyst positions.
Primary Responsibilities:
- Design and execute analytical approaches for large genomic datasets
- Apply statistical and data science methods to genomics problems
- Integrate genomic data with phenotypic or clinical information
- Support research, diagnostics, or precision medicine initiatives
Role Overview
| Category | Details |
|---|---|
| Average Salary (India) | ₹10 – ₹16 LPA (mid-level professionals, depending on experience and organization) |
| What They Do / Nature of Work | Analyze and model genomic data using data science methods to support research, diagnostics, or product development. |
| Senior / Global Level Salary | ₹22 – ₹45+ LPA in India; USD 110,000 – 160,000 annually in global markets. |
| Growth Outlook (Next 5 Years) | Strong growth driven by genomics expansion, precision medicine, and data-driven healthcare. |
| Job Mobility | High opportunities across genomics companies, pharma R&D, biotech firms, diagnostics labs, and healthcare technology organizations. |
| Key Benefits | High-impact analytical role, strong global demand, exposure to advanced genomics and data science, and a pathway to senior scientific or leadership roles. |
- Proteomics Data Analyst-
About the Role: A Proteomics Data Analyst specializes in analyzing protein-level data generated from techniques such as mass spectrometry. This role focuses on identifying proteins, quantifying expression levels, and interpreting proteomic patterns to support research in drug discovery, biomarker identification, and disease mechanism studies. It is an intermediate-level role typically pursued after gaining experience in bioinformatics or omics data analysis.
Primary Responsibilities:
- Analyze proteomics datasets generated from mass spectrometry experiments
- Identify and quantify proteins and peptides
- Interpret proteomic results in biological and experimental contexts
- Collaborate with research and discovery teams to support scientific conclusions.
Role Overview
| Category | Details |
|---|---|
| Average Salary (India) | ₹9 – ₹15 LPA (mid-level professionals, depending on experience and organization) |
| What They Do / Nature of Work | Analyze and interpret proteomics data to support research in drug discovery, biomarker analysis, and disease studies. |
| Senior / Global Level Salary | ₹20 – ₹40+ LPA in India; USD 100,000 – 150,000 annually in global markets. |
| Growth Outlook (Next 5 Years) | Steady growth driven by increasing use of proteomics in drug discovery, biomarker research, and systems biology. |
| Job Mobility | Moderate to high opportunities across pharma R&D, biotech firms, research institutes, and proteomics service providers. |
| Key Benefits | Specialized skill set, strong relevance in drug discovery, exposure to advanced experimental data, and a pathway to senior scientific roles. |
- Translational Bioinformatics Scientist-
About the Role: A Translational Bioinformatics Scientist focuses on bridging biological research data with clinical and real-world applications. This role translates insights from genomics, proteomics, and other omics data into outcomes that support drug development, biomarker discovery, and precision medicine. It is an advanced role that typically comes after strong experience in genomics, computational biology, or data science.
Primary Responsibilities:
- Integrate research-level biological data with clinical or real-world datasets
- Translate multi-omics findings into clinically or therapeutically relevant insights
- Support biomarker identification, patient stratification, and treatment strategies
- Guide analytical approaches for translational and precision-medicine programs
Role Overview
| Category | Details |
|---|---|
| Average Salary (India) | ₹15 – ₹25 LPA (advanced professionals, depending on experience and organization) |
| What They Do / Nature of Work | Translate biological and omics data into clinically meaningful insights that support drug development and precision medicine. |
| Senior / Global Level Salary | ₹30 – ₹50+ LPA in India; USD 120,000 – 180,000 annually in global markets. |
| Growth Outlook (Next 5 Years) | Strong growth driven by precision medicine, translational research, and data-driven clinical development. |
| Job Mobility | High opportunities across pharma R&D, biotech companies, clinical research organizations, and healthcare innovation teams. |
| Key Benefits | High-impact scientific role, strong strategic relevance, global demand, and a pathway to principal or leadership positions. |
- Principal Bioinformatics Scientist-
About the Role: A Principal Bioinformatics Scientist is a senior leadership role responsible for defining bioinformatics strategy, guiding complex analytical programs, and making high-impact scientific decisions. This role oversees advanced bioinformatics initiatives across drug discovery, genomics, translational research, or clinical development, and typically comes after several years of deep technical and domain experience.
Primary Responsibilities:
- Lead and design advanced bioinformatics and multi-omics strategies
- Oversee complex analytical projects across research and clinical programs
- Guide teams on data interpretation, modeling approaches, and best practices
- Collaborate with senior stakeholders across R&D, clinical, and product teams
Role Overview
| Category | Details |
|---|---|
| Average Salary (India) | ₹20 – ₹35 LPA (senior leadership level, varies by organization and responsibility) |
| What They Do / Nature of Work | Define bioinformatics strategy, lead advanced analytics, and guide scientific decision-making across programs. |
| Senior / Global Level Salary | ₹40 – ₹70+ LPA in India; USD 140,000 – 200,000+ annually in global markets. |
| Growth Outlook (Next 5 Years) | Strong demand driven by complex data needs, AI integration, and precision medicine initiatives. |
| Job Mobility | High opportunities across pharma R&D, biotech leadership teams, genomics companies, and global research organizations. |
| Key Benefits | High strategic impact, leadership authority, global recognition, and influence over scientific direction and innovation. |
Clinical Bioinformatics Director –
About the Role: A Clinical Bioinformatics Director (or Clinical Bioinformatics Lead) oversees the application of bioinformatics in clinical and patient-facing contexts. This role ensures that genomic and biological data analysis directly supports diagnostics, treatment decisions, and precision medicine initiatives. It is a senior role typically held by professionals with deep experience in clinical genomics, regulatory environments, and cross-functional leadership.
Primary Responsibilities:
- Lead clinical bioinformatics programs and teams
- Oversee genomic data analysis used for diagnostics and patient care
- Ensure compliance with clinical, regulatory, and data quality standards
- Guide strategy for precision medicine and clinical genomics initiatives
Role Overview
| Category | Details |
|---|---|
| Average Salary (India) | ₹20 – ₹35 LPA (senior clinical leadership level, varies by organization) |
| What They Do / Nature of Work | Lead and govern clinical bioinformatics activities supporting diagnostics, treatment, and precision medicine. |
| Senior / Global Level Salary | ₹40 – ₹70+ LPA in India; USD 140,000 – 200,000+ annually in global markets. |
| Growth Outlook (Next 5 Years) | Strong growth driven by expansion of clinical genomics and precision medicine programs. |
| Job Mobility | Moderate to high opportunities across hospitals, diagnostic labs, pharma, biotech, and healthcare research organizations. |
| Key Benefits | High clinical impact, leadership authority, global relevance, and long-term career stability. |
Career Progression in Bioinformatics
Career growth in bioinformatics is typically driven by a combination of biological knowledge, computational skills, and real-world project experience. Professionals gradually move from executing predefined analysis workflows to designing analytical strategies and leading research initiatives.
As experience increases, responsibilities expand to include interpreting complex biological data, collaborating with multidisciplinary research teams, and guiding scientific decision-making. Senior roles often involve strategic planning, project leadership, and translating biological insights into clinical or commercial applications. The bioinformaticians salary typically increases significantly with experience and specialization.
Bioinformatics Salaries and Career Opportunities in India
India is emerging as a significant hub for bioinformatics due to its expanding biotechnology sector, growing pharmaceutical research ecosystem, and increasing investment in genomics and precision medicine.
Several factors are driving opportunities in India:
- Expansion of genomics and sequencing laboratories
- Growth of pharmaceutical and biotechnology research centers
- Increasing clinical research and drug discovery activities
- Government-supported life science research initiatives
Major cities such as Bangalore, Hyderabad, Pune, Delhi NCR, and Chennai host many biotechnology companies, research institutions, and genomics laboratories that actively hire bioinformatics professionals. In India, bioinformaticians salary ranges from ₹3 LPA at entry level to ₹30+ LPA in advanced roles.
Bioinformatics Career Opportunities and Salaries in Global Markets
Globally, bioinformatics careers are expanding rapidly as healthcare and life sciences become increasingly data-driven, particularly with the growing adoption of genomics and precision medicine in global health systems. Countries such as the United States, Germany, the United Kingdom, Canada, and Singapore have strong demand for bioinformatics professionals in areas like genomics research, drug discovery, and clinical data analysis.
Large pharmaceutical companies, biotechnology firms, genomics startups, and academic research institutions rely heavily on bioinformatics expertise to interpret biological datasets and support scientific innovation.
As precision medicine and AI-enabled biological analysis continue to grow, global demand for bioinformatics professionals is expected to remain strong across research, healthcare, and biotechnology industries. Globally, bioinformaticians’ salaries can exceed USD 100,000 annually for experienced professionals.
Bioinformatics
Develop in-depth skills to analyze, manage, and interpret large-scale biological data used in genomics, clinical research, and drug discovery. This program focuses on applying computational methods and analytical pipelines to transform complex biological data into actionable research insights. The program is designed as practical bioinformatics training aligned with industry workflows.
AI and Automation Trends in Bioinformatics
Artificial intelligence is rapidly becoming part of modern biological research. As a result, bioinformatics professionals are increasingly expected to understand AI-driven analytical approaches. These skills allow researchers to analyze complex biological datasets using advanced computational methods.
Key AI-driven skills include:
– Machine learning applications in genomics and biological data analysis
– Deep learning for biological sequence and structure prediction
– Predictive modeling for drug discovery and biomarker identification
– Handling large-scale omics datasets from public biological databases such as the NCBI genomic repositories
These skills are becoming particularly important in fields such as precision medicine, genomics research, and AI-driven drug discovery.
Conclusion
Bioinformatics has become a critical field in modern life sciences, helping researchers and healthcare organizations interpret large biological datasets. As organizations increasingly rely on biological data to guide decisions, the need for skilled bioinformatics professionals continues to grow across industries and geographies. This makes bioinformatics a stable, future-oriented career choice rather than a short-term trend.
What makes this field especially attractive is its structured career progression from beginner roles that focus on data handling and analysis to advanced positions involving strategy, translation, and leadership. With the right mix of biological understanding, computational skills, and continuous upskilling, professionals can build meaningful and impactful careers in this domain. To gain deeper, job-oriented knowledge and practical exposure, enrolling in a structured bioinformatics course like the Advanced Diploma in Bioinformatics can help you build the skills required to enter and grow in this field with confidence.
Frequently Asked Questions
Physician Assistants use clinical documentation tools such as EHR systems, AI-powered documentation software, voice dictation tools, medical scribes, and clinical reference platforms to manage patient records efficiently. These tools reduce documentation burden, improve accuracy, and ensure compliance in healthcare workflows.
Clinical documentation is essential for care of continuity, legal compliance, insurance billing, and coordinated treatment. However, growing patient loads mkes timely and accurate record-keeping more challenging.
With increasing digital systems, documentation tools now directly affect efficiency and accuracy in clinical workflows. They influence charting time, after-hours work, and decision support within EHR systems.
To manage this, tools like EHR systems, AI-assisted notes, voice dictation, and scribe support are increasingly used to reduce administrative burden while maintaining clinical responsibility.
Professionals interested in building careers in healthcare documentation often pursue a clinical documentation course, medical coding course, or clinical research course to develop industry-relevant skills.
Build practical skills in medical coding and clinical documentation used across hospitals, healthcare providers, and global healthcare services. This medical coding course also functions as a practical clinical documentation course for professionals interested in documentation-driven healthcare roles. Learn how diagnoses, procedures, and services are accurately translated into standardized medical codes for billing, compliance, and data integrity.
Learn More
What is Clinical Documentation in Healthcare?
Clinical documentation refers to the systematic recording of patient health information, including medical history, diagnoses, treatments, procedures, and outcomes. It is a core part of clinical documentation management that ensures accurate and organized clinical documentation across healthcare settings. A well-designed clinical records management system also helps healthcare providers securely store, retrieve, and manage patient information while supporting continuity of care, auditing, and healthcare analytics across departments. This information is stored and managed through a clinical records management system, such as electronic health records (EHRs), to support continuity of care, clinical decision-making, billing accuracy, and legal compliance. Proper documentation improves patient safety, enhances communication among healthcare providers, and ensures all clinical actions are traceable and evidence based. Many professionals strengthen these skills through a clinical documentation course or medical coding course focused on healthcare documentation standards.
Start Clinical Documentation Training
Who is a Physician Assistant and What is their role in Clinical Documentation?
Physician Assistants (PAs) are licensed healthcare professionals who work closely with doctors to support patient care across clinics, hospitals, and emergency settings. They are involved in examining patients, reviewing medical history, ordering diagnostic tests, assisting in diagnosis, and contributing to treatment decisions. In many healthcare environments, PAs serve as a key point of contact for patients, combining clinical responsibility with decision-making support.
As part of their daily responsibilities, Physician Assistants play a direct and active role in clinical documentation. They record patient history, examination findings, assessments, diagnoses, treatment plans, follow-ups, and discharge information in the medical record. This documentation supports continuity of care, enables clear communication across healthcare teams, and meets legal, regulatory, and billing requirements. Accurate documentation by PAs is essential because it reflects real-time clinical decisions and directly influences patient safety and care outcomes.
Types of Clinical Documentation Tools Used by Physician Assistants
Documentation tools influence three key areas of a PA’s daily workflow: efficiency, accuracy, and compliance. Effective clinical documentation management ensures that records are structured, compliant, and aligned with billing and regulatory standards. In many healthcare environments, documentation quality directly influences reimbursement, audit outcomes, and interdepartmental communication. Inaccurate or incomplete records can lead to claim denials, compliance issues, or patient safety concerns. As healthcare systems increasingly rely on structured data for quality reporting and performance metrics, the accuracy of documentation becomes measurable and accountable. For Physician Assistants, this means documentation tools are not merely administrative aids they shape clinical defensibility, workflow efficiency, and professional credibility. The various types of clinical documentation tools used are:
- EHR Systems: Epic Systems, Oracle Cerner, Athenahealth
- AI Documentation Tools: Suki AI, Notable, DeepScribe
- Voice Dictation Tools: Dragon Medical One
- Medical Scribe Solutions: Augmedix, ScribeAmerica
- Clinical Reference Tools: UpToDate, DynaMed, Epocrates
Comparison of Best Clinical Documentation Tools
Clinical documentation tools help healthcare organizations streamline patient record management, improve documentation accuracy, reduce administrative workload, and support efficient clinical workflows. Below is a comparison table of the best clinical documentation tools used across hospitals, clinics, and healthcare systems.
Clinical Documentation Tools Used in Healthcare
| Category | Tool | Primary Function |
|---|---|---|
| EHR System | Epic Systems | Integrated electronic health record and clinical workflow management |
| EHR System | Oracle Cerner | Enterprise-level EHR and interoperability management |
| EHR System | Athenahealth | Cloud-based EHR and outpatient practice management |
| AI Documentation Tool | Suki AI | AI-powered voice-assisted clinical note generation |
| AI Documentation Tool | Notable | AI automation for documentation and patient intake workflows |
| AI Documentation Tool | DeepScribe | Ambient AI medical scribing and clinical note creation |
| Voice Dictation Tool | Dragon Medical One | Real-time speech recognition for medical documentation |
| Medical Scribe Solution | Augmedix | Remote medical scribing and documentation support |
| Medical Scribe Solution | ScribeAmerica | Human medical scribe services for healthcare providers |
| Clinical Reference Tool | UpToDate | Evidence-based clinical decision support resource |
| Clinical Reference Tool | DynaMed | Point-of-care clinical reference and treatment guidance |
| Clinical Reference Tool | Epocrates | Drug reference and clinical information support tool |
Tools Used by Physician Assistants for Clinical Documentation Support
Basic Documentation Tools (Foundational Systems)
Basic documentation tools form the foundation of a Physician Assistant’s daily workflow. These systems are used to document patient encounters, manage records, reference clinical guidelines, and ensure regulatory compliance. Proficiency in these platforms is considered essential for safe, structured, and legally sound documentation across healthcare settings. These platforms collectively function as a clinical records management system, allowing healthcare organizations to store, retrieve, and standardize patient documentation across departments.
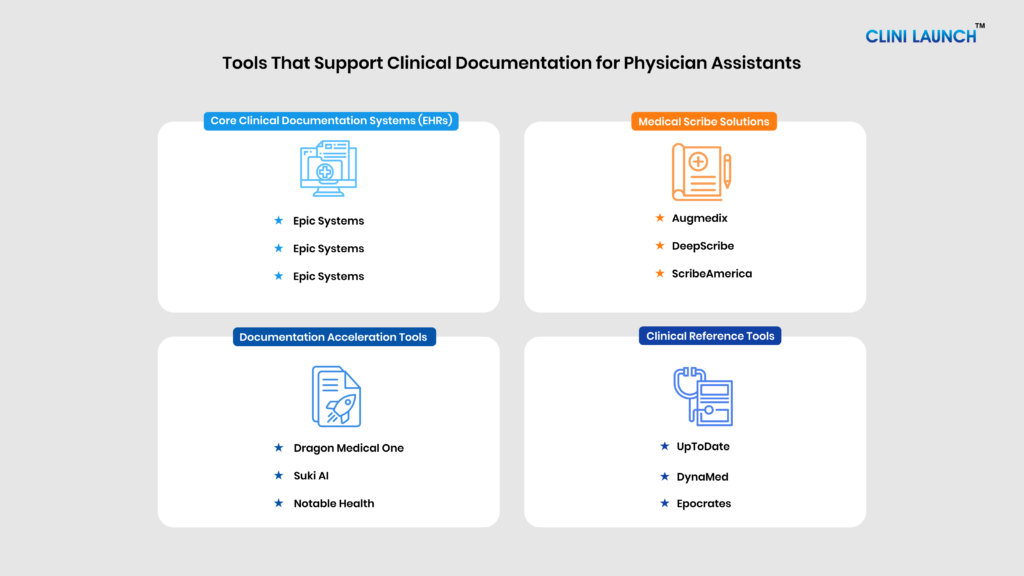
1. Epic Systems
Epic is a comprehensive enterprise-level Electronic Health Record (EHR) system widely used in large hospitals and academic medical centers. It enables Physician Assistants to document complete patient encounters, manage orders, review labs and imaging, and support billing-compliant workflows within a structured environment. Because Epic is deeply integrated into hospital systems, proficiency in navigating its templates and documentation standards is considered a foundational competency for PAs working in inpatient and high-volume care settings.
2. Cerner (Oracle Health)
Cerner, now part of Oracle Health, is an enterprise EHR platform commonly used in emergency departments and acute care environments. It supports real-time documentation, order entry, medication management, and interdisciplinary communication within fast-paced clinical workflows. For Physician Assistants working in hospital-based roles, familiarity with Cerner helps improve efficiency under time pressure and supports accurate, workflow-aligned documentation.
3. Athenahealth
Athenahealth is a cloud-based EHR platform primarily used in outpatient and ambulatory care settings. It allows Physician Assistants to document patient visits, manage prescriptions, handle billing tasks, and coordinate follow-ups within a streamlined interface. Its simplified design supports quicker documentation turnaround, making it especially useful for PAs managing high patient throughput in clinic environments.
4. UpToDate
UpToDate is an evidence-based clinical decision support resource widely used across healthcare institutions. While it does not generate documentation, it supports Physician Assistants by strengthening the clinical reasoning and evidence behind diagnoses, treatment plans, and management decisions recorded in patient notes. Its regularly updated content helps ensure documentation aligns with current medical standards.
5. DynaMed
DynaMed is a point-of-care clinical reference tool that provides concise, structured summaries of medical conditions and treatment guidelines. It supports rapid clinical confirmation during patient encounters, helping Physician Assistants document care decisions accurately and efficiently. Its streamlined format is particularly useful in time-sensitive clinical environments.
6. Epocrates
Epocrates is a widely used drug reference tool that provides information on medication dosing, interactions, contraindications, and safety considerations. While not a documentation system itself, it supports accurate medication-related entries within patient records. For Physician Assistants managing prescribing responsibilities, it strengthens documentation reliability and reduces the risk of medication errors.
AI in Clinical Documentation & Automation Trends for PAs
Advanced documentation tools focus on accelerating workflows through voice recognition, AI-assisted drafting, and scribe support. These technologies reduce manual charting burden but still require active review and approval by the Physician Assistant. Understanding how these tools function is increasingly important in digitally integrated healthcare systems.
1. Nuance Dragon Medical One
Dragon Medical One is a medical-grade voice recognition tool that converts spoken clinical notes into structured documentation within the EHR. It integrates with major platforms such as Epic and Cerner, enabling real-time dictation during patient encounters. For Physician Assistants managing busy clinics or ward rounds, voice-enabled documentation significantly reduces typing time while maintaining note accuracy and completeness.
2. Suki AI
Suki AI is an AI-powered clinical documentation assistant designed to help clinicians generate structured SOAP notes using voice commands or conversational inputs. It drafts documentation within the EHR, allowing Physician Assistants to review, edit, and finalize notes before submission. By reducing repetitive formatting and data entry, Suki supports faster documentation completion while maintaining clinical oversight and responsibility.
3. Notable Health
Notable Health uses AI-driven automation to assist with documentation and post-visit workflows, including follow-ups and administrative tasks. Rather than replacing clinical input, it streamlines repetitive documentation processes that often extend beyond patient encounters. For Physician Assistants in high-efficiency outpatient settings, this reduces after-hours charting and improves overall workflow balance.
Medical Scribe Solutions are designed to reduce how much documentation a Physician Assistant needs to personally write. In this model, a human scribe, AI system, or a combination of both prepares the clinical note, which the PA then reviews, edits if needed, and signs off.
4. Augmedix
Augmedix provides remote medical scribe services supported by AI, where trained professionals prepare clinical documentation based on patient encounters. The Physician Assistant reviews and approves the drafted notes before final submission. This hybrid model significantly reduces the time spent writing detailed notes, especially in high-volume clinical environments, while preserving clinician accountability.
5. DeepScribe
DeepScribe uses ambient AI technology to listen during patient visits and automatically generate structured clinical notes without requiring active dictation. The system operates in the background and produces draft documentation for review. For Physician Assistants seeking minimal workflow interruption during patient interaction, this hands-free model supports documentation efficiency while maintaining final clinical oversight.
6. ScribeAmerica
ScribeAmerica provides trained human medical scribes who document patient encounters directly within the EHR under clinician supervision. This model significantly reduces the amount of manual charting required from Physician Assistants, particularly in emergency departments and inpatient settings. The PA remains responsible for reviewing and signing off on documentation to ensure accuracy and compliance.
How Documentation Tools Influence Burnout and Workload
Administrative workload is one of the most cited contributors to clinician burnout. For Physician Assistants managing high patient volumes, after-hours charting can extend the clinical day significantly. Documentation acceleration tools, ambient AI systems, and scribe support reduce time spent typing and formatting notes. However, efficiency gains depend on proper tool selection and workflow integration. Tools that require excessive correction or poor EHR integration may increase frustration rather than reduce it. The true value of documentation systems lies not just in speed, but in how seamlessly they fit into real-world clinical routines.
Competencies Required for Physician Assistants
Beyond technical familiarity, PAs must develop structured thinking in documentation. This includes understanding how to write defensible clinical notes, documenting medical decision-making clearly, and aligning documentation with billing levels of service. As AI-generated drafts become more common, the ability to detect subtle inaccuracies, missing context, or incorrect auto-populated data becomes critical. Competency now extends beyond writing — it includes reviewing, validating, and ensuring compliance before final sign-off. To use modern documentation tools effectively, Physician Assistants must develop structured documentation skills, EHR navigation proficiency, and awareness of billing-compliant note formatting. Advanced AI and scribe tools also require the ability to critically review auto-generated content, correct inaccuracies, and ensure clinical responsibility remains intact. In specialized roles focused on documentation accuracy and reimbursement compliance, professionals may also pursue CDI certification training to develop expertise in clinical documentation improvement processes.
For professionals interested in expanding into documentation-driven healthcare roles such as clinical research, regulatory affairs, medical coding, or positions like a clinical documentation specialist, structured training becomes increasingly valuable. A clinical documentation course helps learners understand healthcare workflows and documentation standards, while a medical coding course develops expertise in coding systems, reimbursement processes, and compliance. A clinical research course further supports professionals interested in protocol documentation, audit readiness, clinical trial documentation, and regulatory operations.
Medical Coding
Build practical skills in medical coding and clinical documentation used across hospitals, healthcare providers, and global healthcare services. This program also functions as a practical clinical documentation course for professionals interested in documentation-driven healthcare roles. Learn how diagnoses, procedures, and services are accurately translated into standardized medical codes for billing, compliance, and data integrity.
As healthcare shifts toward federally supported digital health infrastructure and Electronic Health Record (EHR) standards, tool proficiency becomes a professional competency — not just a technical skill.
Clinical Documentation Specialist Career Opportunities
The demand for skilled clinical documentation specialist professionals is increasing across hospitals, healthcare BPOs, medical coding companies, insurance firms, and healthcare technology organizations. With the growing adoption of digital healthcare systems and clinical records management platforms, professionals trained through a clinical documentation course can explore roles related to medical documentation review, healthcare compliance, clinical data quality, and coding support. Learners from a medical coding course often develop expertise in documentation accuracy, coding systems, and reimbursement workflows, while professionals from a clinical research course gain exposure to protocol documentation, regulatory compliance, audit readiness, and healthcare data management. Many learners also pursue cdi certification training to strengthen their expertise, improve career prospects, and qualify for advanced opportunities in the healthcare documentation and revenue cycle management industry.
Clinical Documentation Specialist Salary in India
The salary of a Clinical Documentation Specialist in India varies based on experience, healthcare domain knowledge, certifications, and familiarity with clinical documentation software and healthcare compliance standards. Freshers entering the field after completing a clinical documentation course or medical coding course can expect competitive entry-level salaries, while experienced professionals working with hospitals, healthcare BPOs, medical coding companies, and clinical documentation management teams often earn significantly higher packages. Professionals trained through a clinical research course may also transition into documentation-focused healthcare and regulatory roles. Expertise in medical terminology, clinical records management systems, ICD coding, and healthcare analytics continues to increase demand across the healthcare industry.
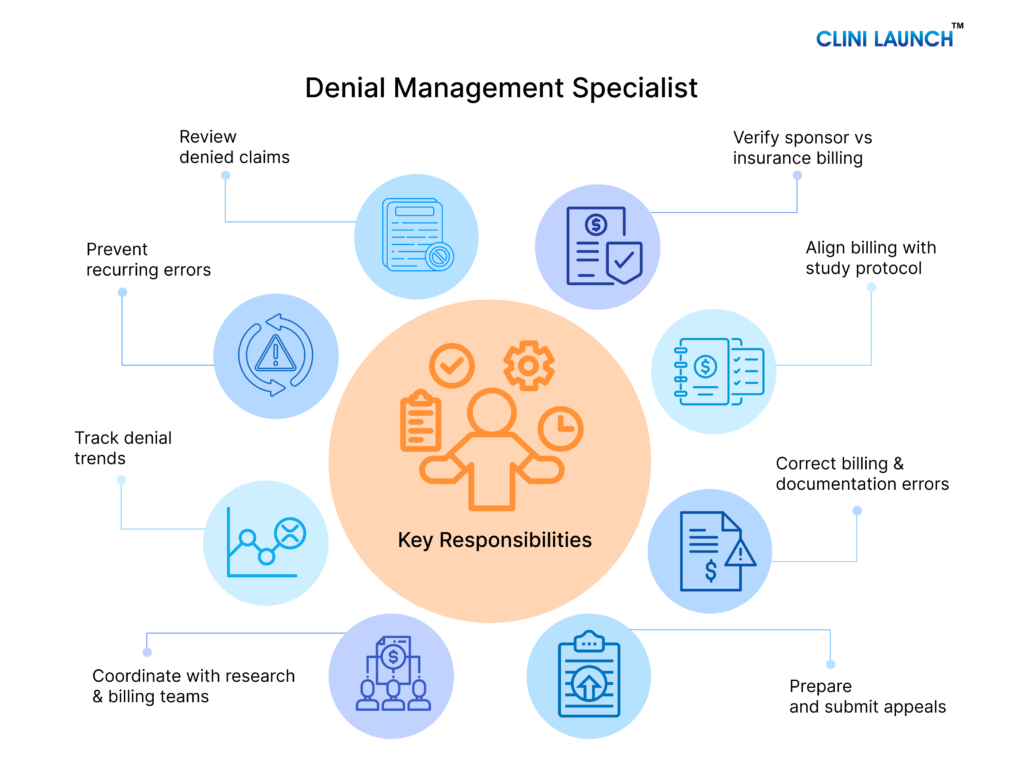
Salary Range for Denial Management Roles in India
| Level | Average Salary Range in India | Experience |
|---|---|---|
| Entry Level | ₹2.5 LPA – ₹4.5 LPA | 0 – 2 Years |
| Mid-Level | ₹5 LPA – ₹8 LPA | 3 – 6 Years |
| Senior Level | ₹9 LPA – ₹15+ LPA | 7+ Years |
Clinical Research
Build industry-ready skills to work across real clinical trial environments. Learn how clinical studies are designed, conducted, documented, and monitored, with a strong focus on ethics, patient safety, and global regulatory compliance.
Workflow Impact of Documentation Tools
Documentation tools influence not only how notes are written, but also when and where Physician Assistants complete them. The comparison below highlights how different systems affect after-hours of charting, real-time documentation, and adaptability to clinical workflow.
Workflow Impact Comparison
| Tool | When Documentation Happens | Effect on After-Hours Charting | Adaptability to PA Workflow |
|---|---|---|---|
| Epic Systems | During & after visit | Moderate | High |
| Cerner (Oracle Health) | During & after visit | Moderate | High |
| athenahealth | Mostly during visit | Low | Moderate |
| Nuance Dragon Medical One | During visit | Low | High |
| Suki AI | During visit | Low | Moderate |
| Notable Health | After visit | Very Low | Moderate |
| Augmedix | During visit | Very Low | Moderate |
| DeepScribe | During visit | Very Low | Low–Moderate |
| ScribeAmerica | During visit | Very Low | Low |
| UpToDate | During documentation | N/A | High |
| DynaMed | During documentation | N/A | High |
| Epocrates | During prescribing | N/A | High |
Risks and Limitations of Clinical Documentation Tools
While documentation technologies improve efficiency, they introduce new responsibilities. AI-generated drafts may contain subtle errors. Ambient transcription systems may misinterpret clinical nuance. Overreliance on structured templates may oversimplify complex patient cases. Understanding these risks ensures that efficiency does not compromise clinical judgment.
• Overreliance on AI-generated documentation
• Risk of inaccuracies in ambient transcription
• Patient privacy concerns in audio-recorded encounters
• Data security risks
• Alert fatigue within EHR systems
• Reduced critical thinking if documentation is blindly accepted
While tools enhance efficiency, final accountability always remains with the Physician Assistant.
The Future of Clinical Documentation for PAs
• Ambient AI documentation becoming more common
• Real-time coding integration within notes
• Predictive documentation prompts
• Greater automation of billing alignment
• Increased AI-assisted compliance checks
The future of documentation will likely be hybrid combining human clinical reasoning with intelligent automation.
Conclusion
Clinical documentation has become one of the most critical and time-intensive responsibilities in a Physician Assistant’s role. Effective clinical documentation management now supports continuity of care, regulatory compliance, billing accuracy, and audit readiness across healthcare systems. Beyond recording patient encounters, documentation today supports continuity of care, regulatory compliance, billing accuracy, audits, and data-driven decision-making across healthcare systems. As digital platforms, AI-assisted tools, and automation become standard, understanding how documentation works within these systems is no longer optional.
From core EHR platforms to documentation acceleration tools, scribe solutions, and clinical reference systems, each category plays a distinct role in shaping how Physician Assistants manage their workload and maintain clinical accuracy. The ability to work effectively with these tools depends not only on technology, but also on a strong understanding of clinical workflows, compliance requirements, and structured healthcare documentation.
The evolution of documentation tools reflects a broader shift in healthcare from manual record-keeping to structured, data-driven systems. As digital infrastructure expands, Physician Assistants who understand both clinical reasoning and documentation systems will be better positioned to adapt to evolving regulatory and technological environments.
For professionals interested in building careers around clinical documentation, regulatory processes, and healthcare data beyond bedside roles formal training becomes increasingly important. Clinilaunch Research Institute offers clinical research course that equips learners with practical knowledge of clinical documentation, protocols, audits, and data handling used across hospitals, CROs, and research organizations, helping them transition confidently into documentation-driven healthcare roles. Professionals can also strengthen their expertise through a clinical documentation course or medical coding course focused on healthcare documentation standards, coding systems, and compliance workflows.