The best biomedical research tools used in the industry include PCR and qPCR systems for diagnostics, LIMS for laboratory workflow management, BLAST for bioinformatics, R, Python, and SAS for data analysis, Medidata Rave for clinical trials, eTMF for documentation, Argus Safety for pharmacovigilance, and EHR systems for healthcare records. These tools support diagnostics, clinical research, drug safety, and healthcare operations, forming the foundation of modern biomedical careers.
The biomedical industry is not driven by theory alone. It depends on the best biomedical research tools that support diagnostics, research, clinical studies, and healthcare data workflows. Core examples include PCR and qPCR systems for molecular diagnostics, LIMS for laboratory data management, bioinformatics tools like BLAST, statistical platforms such as R, Python, and SAS, clinical trial systems like Medidata Rave, documentation platforms such as eTMF systems, pharmacovigilance tools like Argus Safety, and Electronic Health Record (EHR) platforms used in healthcare.
Knowing which tools matter is often more valuable than trying to learn everything at once, especially when identifying the best biomedical research tools used in real-world settings. One trend that underscores this shift is the rapid growth of clinical data analytics tools, with the global market expected to grow from around US $18.8 billion in 2023 to over US $188 billion by 2033, highlighting how analytics, EHR systems, and digital platforms are reshaping biomedical workflows.
This blog breaks down the best biomedical research tools that every graduate should be aware of. From medical laboratory tools and diagnostic tools in biomedical research to digital platforms supporting clinical research tools, safety, quality, and healthcare records, these tools form the foundation of modern biomedical careers.
Bioinformatics
Build practical skills to analyze and interpret biological data generated from genomics, proteomics, and clinical research studies. Learn how computational tools and data-driven methods are used to convert raw biological data into meaningful insights for research and drug development.

Duration: 6 months
Learn at your own pace
Skills you’ll build:
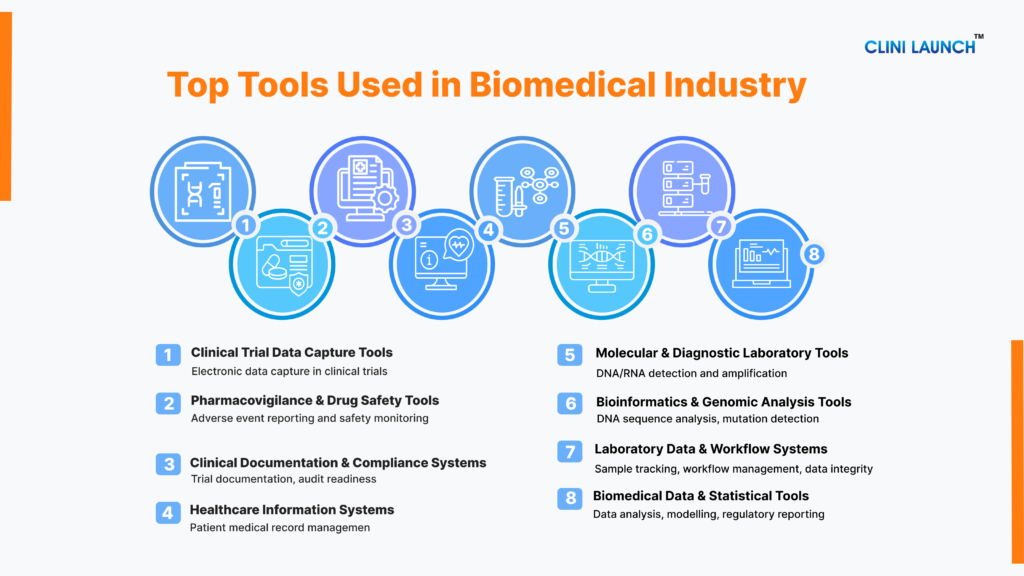
A. Molecular & Diagnostic Laboratory Tools
Molecular and diagnostic laboratory tools form the foundation of biomedical science by generating primary biological data from patient samples such as blood, swabs, or tissue. The accuracy of this data directly impacts downstream processes including clinical research, drug safety monitoring, and healthcare analytics.
In practice, these tools are used for detecting pathogens, identifying genetic variations, and supporting diagnostic decision-making. Among the most essential tools in this category are PCR and qPCR systems.
1. PCR (Polymerase Chain Reaction)
Kary Mullis developed PCR, a technique used to amplify specific DNA sequences from very small samples, enabling detection even at extremely low concentrations.
It is widely used to:
- Detect infectious diseases
- Identify genetic mutations
- Support research and forensic analysis
2. qPCR (Quantitative PCR)
qPCR, or real-time PCR, builds on PCR by measuring DNA amplification as it occurs using fluorescence signals, allowing both detection and quantification of genetic material.
It is used to:
- Measure viral load in infections
- Analyse gene expression
- Track biomarkers in drug development
B. Laboratory Data & Workflow Management Systems
While molecular tools generate biological data, laboratory data systems ensure that this data is structured, traceable, and compliant. In modern biomedical environments, data integrity is as critical as scientific accuracy.
Two key systems in this category are LIMS and ELN platforms.
1. LIMS (Laboratory Information Management System)
A LIMS acts as the operational backbone of laboratories by managing samples, workflows, and reporting processes. It ensures complete traceability from sample collection to final output.
It is widely used for sample tracking, automation of workflows, audit trails, and compliance reporting in diagnostic labs and pharmaceutical environments.
2. ELN (Electronic Lab Notebook)
An ELN replaces paper-based documentation with structured digital recording of experiments and observations. While LIMS manages operations, ELN focuses on experimental documentation and collaboration.
It is commonly used in research and R&D environments to improve transparency, prevent data duplication, and support regulatory compliance.
C. Bioinformatics & Genomic Analysis Tools
Bioinformatics and genomic analysis tools transform raw DNA and RNA data into meaningful insights used in research, diagnostics, and precision medicine. As genomics advances, these tools are becoming essential for understanding genetic variations and disease mechanisms.
In practice, they are used for sequence comparison, variant detection, and workflow-based genomic analysis. Among the most widely used tools are BLAST, GATK, and Galaxy.
1. BLAST (Basic Local Alignment Search Tool)
BLAST is used to compare DNA and protein sequences against large biological databases to identify similarities, functions, and mutations.
It is commonly used to:
- Identify genes and sequence similarities
- Analyse mutations and variations
- Validate sequencing results
2. GATK (Genome Analysis Toolkit)
GATK is a widely used framework for analysing high-throughput sequencing data.
It is used to:
- Detect genetic variants (SNPs, insertions, deletions)
- Process large-scale genomic datasets
- Support clinical genomics and precision medicine research
3. Galaxy
Galaxy is a user-friendly platform that enables genomic analysis without requiring programming skills.
It is used to:
- Perform workflow-based sequence analysis
- Run RNA-seq and variant analysis pipelines
- Support reproducible research in academic settings
D. Biomedical Data & Statistical Analysis Tools
Once biomedical data is generated, statistical tools convert it into meaningful insights that support clinical decisions, research outcomes, and regulatory submissions. These tools are widely used across clinical trials, healthcare analytics, and biomedical research.
In practice, they support data cleaning, statistical modelling, visualization, and preparation of datasets for reporting and analysis.
1. R
R is widely used for statistical modelling, data analysis, and visualization in biomedical research and biostatistics.
It is commonly used to:
- Perform statistical analysis and modelling
- Analyse clinical and genomic datasets
- Generate data visualizations and reports
2. Python
Python is a versatile programming language used for data processing, machine learning, and AI-driven healthcare applications.
It is used to:
- Process and analyse large datasets
- Build predictive models and automation workflows
- Support AI and machine learning in healthcare
3. SAS
SAS is the industry-standard tool for clinical trial analysis and regulatory reporting.
It is used to:
- Analyse clinical trial data
- Generate Tables, Listings, and Figures (TLFs)
- Prepare submission-ready datasets for regulatory authorities
E. Clinical Trial Data Capture Tools
Clinical trial data capture tools ensure that patient data is collected, validated, and maintained in a structured and regulatory-compliant manner across study sites. In global trials, these systems are essential for maintaining data accuracy, consistency, and traceability.
In practice, they support workflows such as electronic Case Report Form (eCRF) design, data entry, validation checks, query management, and preparation of datasets for statistical analysis.
1. Medidata Rave
Medidata Rave is a widely used Electronic Data Capture (EDC) system in global clinical trials.
It is used to:
- Design eCRFs and capture patient data
- Validate data and manage queries
- Enable real-time monitoring and audit trails
- Prepare data for downstream analysis
2. Oracle Clinical
Oracle Clinical is used for managing large-scale clinical trial data.
It supports:
- Structured data capture and validation
- Discrepancy and query management
- Integration with other clinical systems
3. REDCap
REDCap is a flexible, web-based tool widely used in academic and investigator-led studies.
It is used to:
- Create custom data collection forms
- Manage research data and participants
- Support smaller or observational studies
F. Clinical Trial Data Capture Tools
Clinical trial data capture tools ensure that patient data is collected, validated, and maintained in a structured and regulatory-compliant manner across study sites. In global trials, these systems are essential for maintaining data accuracy, consistency, and traceability.
In practice, they support workflows such as electronic Case Report Form (eCRF) design, data entry, validation checks, query management, and preparation of datasets for statistical analysis.
1. Medidata Rave
Medidata Rave is a widely used Electronic Data Capture (EDC) platform in global clinical trials.
It is used to:
- Design eCRFs and capture patient data
- Validate data and manage queries
- Maintain audit trails for compliance
- Enable data export for analysis
2. Oracle Clinical
Oracle Clinical is used for managing large-scale clinical trial data.
It supports:
- Structured data capture and validation
- Discrepancy management
- Integration with other clinical systems
3. REDCap
REDCap is a web-based tool commonly used in academic and research settings.
It is used to:
- Create custom data collection forms
- Manage research data
- Support smaller or investigator-led studies
G. Pharmacovigilance & Drug Safety Tools
Pharmacovigilance tools monitor drug safety after products reach patients, ensuring that adverse events are captured, evaluated, and reported in compliance with global regulatory requirements. These systems play a critical role in both clinical trials and post-marketing surveillance, where real-world data can reveal risks not identified during controlled studies.
In practice, pharmacovigilance workflows involve case intake, medical review, coding, signal detection, and regulatory submission—all of which are managed through specialized safety databases.
1. Oracle Argus Safety
Oracle Argus Safety is one of the most widely used pharmacovigilance platforms globally for managing Individual Case Safety Reports (ICSRs).
In real-world operations, Argus Safety is used to:
- Capture and validate adverse event data
- Perform medical coding using standardized dictionaries such as MedDRA
- Generate expedited and periodic safety reports
- Track case workflows and regulatory submission timelines
It is extensively used by pharmaceutical companies and CROs to ensure compliance with global authorities such as the FDA and European Medicines Agency.
2. LifeSphere Safety
LifeSphere Safety is a modern pharmacovigilance platform designed to manage end-to-end drug safety workflows across global operations.
In industry use, LifeSphere Safety supports:
- Case intake and processing across multiple sources
- Signal detection and risk evaluation
- Aggregate reporting and benefit-risk assessment
- Integration with regulatory submission systems
It is particularly valuable for organizations managing large volumes of safety data across multiple regions, helping standardize workflows while maintaining compliance with regional regulations.
Quick Overview of Biomedical Tools & Their Roles
The table below summarizes the key tools used across different areas of the biomedical industry and their practical applications.
Top 15 Biomedical Research Tools Used in Industry
Biomedical Industry Tools
| Category | Tools | What It Does | Where It’s Used |
|---|---|---|---|
| Molecular & Diagnostic Laboratory Tools | PCR / qPCR | Detects and quantifies DNA/RNA | Diagnostics, molecular labs |
| Laboratory Data & Workflow Management Systems | LIMS | Tracks samples and manages lab workflows | Diagnostic labs, pharma labs |
| Laboratory Data & Workflow Management Systems | ELN | Records experiments and research data | R&D, research labs |
| Bioinformatics & Genomic Analysis Tools | BLAST | Compares DNA/protein sequences | Genomics, bioinformatics |
| Bioinformatics & Genomic Analysis Tools | GATK | Analyses sequencing data and genetic variants | Genomics research |
| Bioinformatics & Genomic Analysis Tools | Galaxy | Enables workflow-based genomic analysis | Research, academic labs |
| Biomedical Data & Statistical Analysis Tools | R | Performs statistical analysis and visualization | Research, biostatistics |
| Biomedical Data & Statistical Analysis Tools | Python | Supports data processing, AI, and automation | Healthcare analytics, AI |
| Biomedical Data & Statistical Analysis Tools | SAS | Handles clinical trial analysis and reporting | Clinical research, pharma |
| Clinical Trial Data Capture Tools | Medidata Rave | Captures and manages clinical trial data | Clinical trials (industry) |
| Clinical Documentation & Compliance Systems | eTMF | Stores and manages trial documentation | Regulatory compliance |
| Pharmacovigilance & Drug Safety Tools | Argus Safety | Tracks and reports adverse drug events | Drug safety, pharmacovigilance |
While all these tools are used in the biomedical industry, beginners should prioritize learning PCR, LIMS, BLAST, and basic R/Python, while clinical research roles require expertise in SAS, EDC systems, and eTMF platforms.
Clinical SAS Professional Certificate
Build industry-ready skills in clinical data analysis using SAS, the gold-standard statistical tool used in global clinical trials and regulatory submissions. Learn how clinical trial data is structured, analysed, and reported in compliance with regulatory guidelines while working on real-world clinical datasets and workflows used by pharmaceutical companies and CROs.

Duration: 6 months
Learn at your own pace
Skills you’ll build:
Challenges and Limitations
Despite rapid advancement, biomedical tools face persistent operational and structural challenges. One major issue is data fragmentation. Laboratory systems, clinical trial platforms, pharmacovigilance databases, and hospital information systems often function independently, making integration complex and time-consuming. Poor interoperability increases manual reconciliation efforts and can introduce compliance risks.
Regulatory requirements also slow implementation. Systems must comply with frameworks such as GCP, GMP, and global data protection laws. Every update requires validation and documentation, increasing costs, and reducing agility. Financial barriers further limit adoption, as enterprise-level platforms and advanced laboratory tools demand substantial investment in licensing, infrastructure, cybersecurity, and maintenance.
Workforce readiness remains a constraint. Many tools require specialized laboratory skills, statistical programming expertise, or regulatory knowledge that is not always covered in academic training. Additionally, cybersecurity risks are growing as more platforms move to cloud-based environments, making protection of genomic and patient data a critical concern.
Biomedical Tools Linked to High-Paying Careers
Understanding tools is important—but knowing which tools lead to high-growth, high-paying roles is what matters for career decisions.
Different tools are directly linked to specific job roles across clinical research, data science, pharmacovigilance, and bioinformatics.
Tool → Career Role → Salary Potential
| Tool | Common Job Roles | Salary Potential (India) |
|---|---|---|
| SAS | Clinical Data Analyst, Biostatistician | ₹6–18 LPA |
| Python | Data Scientist, Healthcare AI Specialist | ₹6–25 LPA |
| R | Biostatistician, Research Analyst | ₹5–15 LPA |
| Medidata Rave | Clinical Data Manager, Clinical Trial Associate | ₹5–14 LPA |
| Oracle Clinical | Clinical Data Manager | ₹6–15 LPA |
| Argus Safety | Drug Safety Associate, PV Specialist | ₹4–12 LPA |
| LifeSphere Safety | Pharmacovigilance Specialist | ₹5–14 LPA |
| BLAST / GATK | Bioinformatics Analyst, Genomics Scientist | ₹5–18 LPA |
| LIMS | Laboratory Analyst, Quality Analyst | ₹3–10 LPA |
| PCR / qPCR | Lab Technician, Research Associate | ₹3–8 LPA |
Turn Skills Into High-Paying Career Opportunities
Master clinical SAS, bioinformatics, pharmacovigilance, and clinical research tools that employers actively look for in pharma and healthcare industries.
What This Means to your Career
- Data tools (Python, SAS, R) → Highest salary growth due to analytics + AI demand
- Clinical tools (EDC, SAS) → Strong demand in pharma & CROs
- Bioinformatics tools → High growth in genomics & precision medicine
- Lab tools (PCR, LIMS) → Entry-level but essential foundation
Future Trends
The biomedical tool ecosystem is moving toward deeper integration, automation, and intelligent decision support. Over the next decade, laboratory systems, clinical research platforms, and healthcare technologies are expected to function as interconnected digital environments rather than isolated tools.
AI-native integration is one of the most significant shifts. Artificial intelligence is being embedded directly into statistical platforms, EDC systems, pharmacovigilance databases, and EHR systems to detect anomalies, predict patient risk, and optimize workflows in real time rather than after data collection.
Cloud-connected infrastructures are expanding across LIMS, clinical trial systems, and hospital platforms, enabling remote monitoring, global collaboration, scalable storage, and faster regulatory submissions. This transition reduces local infrastructure dependency while improving operational flexibility.
Precision medicine strengthens the link between molecular diagnostics, bioinformatics, and clinical analytics. Genetic testing data is increasingly integrated with clinical systems to support personalized treatment decisions based on biomarkers and real-world outcomes.
Automation is streamlining validation, coding, reporting, and monitoring processes across clinical and healthcare operations. At the same time, interoperability standards are improving communication between EHR systems, laboratory databases, and regulatory platforms, reducing duplication and enhancing coordinated care.
Digital health technologies such as wearables and remote monitoring tools are also expanding the volume of real-world data available for research and patient management.
Together, these trends are shaping a connected, data-driven biomedical ecosystem where diagnostics, research, safety monitoring, and patient care operate within a unified digital framework.
Ready to Work with Real Biomedical Industry Tools?
Get hands-on exposure to clinical trials, safety reporting, healthcare analytics, and biomedical workflows through career-focused professional programs.
Conclusion
In a biomedical industry shaped by data, diagnostics, and regulatory standards, understanding how the best biomedical research tools are used in real workflows makes a meaningful difference. Whether your interest lies in laboratory operations, clinical data, pharmacovigilance, or healthcare analytics, familiarity with these tools helps bridge the gap between academics and industry expectations.
At CliniLaunch Research Institute, training programs focus on practical exposure to many of the tools used in the biomedical industry, including clinical data workflows, electronic data capture, safety reporting, and quality systems, helping learners understand how these tools are applied in real clinical research environments.