| Clinical Project Associate |
|---|
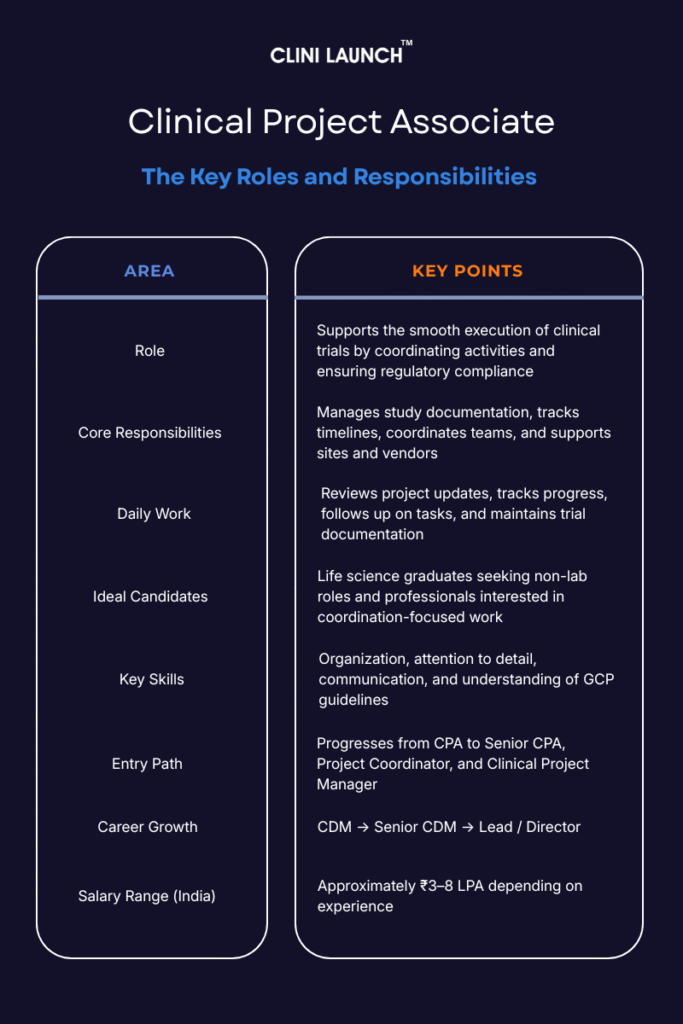
| A Clinical Project Associate (CPA) supports clinical trial operations by managing study documentation, coordinating project activities, supporting sites and vendors, and ensuring compliance with GCP and regulatory standards. The role serves as an entry point into clinical research operations and project management. |
A Clinical Project Associate (CPA) supports the execution of clinical trials by assisting Clinical Project Managers with core operational tasks. The role primarily involves managing study documentation, tracking project timelines, coordinating activities across sites and teams, and supporting regulatory compliance throughout the clinical trial lifecycle.
CPAs work across multiple phases of a clinical trial and support routine operational workflows that help maintain alignment with planned study processes. Their involvement spans different trial stages, ensuring continuity across ongoing activities.
Over the past two decades, clinical research roles have become more complex, with greater emphasis on documentation control, timeline tracking, and regulatory compliance. These evolving demands closely align with the core responsibilities of Clinical Project Associates.
A Clinical Project Associate (CPA) supports clinical trial operations by managing study documentation, coordinating project activities, supporting sites and vendors, and ensuring compliance with GCP and regulatory standards. The role serves as an entry point into clinical research operations and project management.
Who Is a Clinical Project Associate (CPA)?
A Clinical Project Associate is a central operational professional within the clinical trial team who ensures consistency, coordination, and control across trial activities. CPAs act as a link between project managers, clinical sites, vendors, and internal teams, enabling smooth communication and task execution.
The importance of the CPA role lies in its ability to prevent delays, reduce errors, and maintain audit readiness. By keeping documentation accurate, timelines visible, and communication aligned, CPAs help ensure that clinical trials move efficiently from study start-up to close-out, supporting the timely development of new treatments.
CPA vs CRA: Understanding the Difference
While both roles support clinical trials, Clinical Project Associates (CPAs) and Clinical Research Associates (CRAs) have different responsibilities. A CPA focuses on project coordination, documentation, timeline tracking, and operational support, working closely with project managers, sites, and vendors to keep studies organized and compliant.
In contrast, a CRA is primarily responsible for site monitoring activities, including site visits, source data verification, and ensuring protocol adherence at investigational sites. CRAs work directly with trial sites, while CPAs operate more from a central, coordination-based role.
In simple terms, CPAs manage trial operations from behind the scenes, while CRAs oversee trial conduct at the site level.
Core Roles and Responsibilities of a CPA
A CPA is responsible for supporting the coordination and smooth execution of clinical trials, ensuring that operational tasks are completed efficiently, on schedule and in compliance with regulatory standards. CPA roles and responsibilities include supporting documentation, timelines, coordination, and compliance. Below are the core roles and responsibilities of a CPA:
1.Clinical Trial Documentation Oversight
Clinical trials generate large volumes of documents, and even minor documentation errors can delay approvals or trigger audit findings. CPAs are responsible for ensuring that every document is accurate, complete, current, and compliant with regulatory standards.
They help prepare, organize, and maintain essential study documents such as protocols, informed consent forms, investigator brochures, and regulatory submissions. By properly indexing and maintaining study master files, CPAs make sure that documents are easily retrievable during audits, inspections, or sponsor reviews. These clinical project associate duties form the foundation of trial documentation control. This process ensures effective project documentation management throughout the study.
| Task | Explanation |
|---|---|
| Ensure Regulatory Compliance of Documents | CPAs ensure clinical trial documents are accurate, complete, and compliant with GCP and regulatory standards. |
| Prepare and Organize Study Files | CPAs prepare and organize study documents (e.g., protocols, consent forms) and ensure proper indexing and storage for easy audit access. |
| Trial Master File (TMF) Maintenance | CPAs organize and index study documents to maintain the TMF and ensure readiness for audits and inspections. |
2. Project Coordination and Timeline Management
Clinical trials run on strict timelines, and delays at any stage can impact costs, patient recruitment, and regulatory submissions. CPAs support project managers by tracking milestones, monitoring progress, and flagging risks early.
They coordinate activities across teams, help organize meetings, and ensure that assigned tasks are completed as planned. When delays occur, CPAs assist in identifying the cause and work with the team to adjust timelines or re-prioritize activities.
| Task | Explanation |
|---|---|
| Track Project Progress | CPAs ensure project milestones are achieved on time and assist in adjusting plans if there are delays. |
| Monitor Project Timelines | CPAs ensure tasks remain on schedule and adjust timelines when necessary to meet project goals. |
| Coordinate Team Activities | CPAs organize team meetings and ensure activities align with project objectives and deadlines, keeping all team members on track. |
| Identify and Address Delays | CPAs proactively identify delays and collaborate with the team to resolve issues quickly. |
3. Site Operations & External Partner Coordination
Clinical trial sites and vendors play a crucial role in study execution, and CPAs support their readiness and compliance. During site initiation, CPAs help ensure that all required documents, supplies, and systems are in place before patient enrolment begins. Vendor coordination is a core responsibility during trial execution.
They also assist in tracking site performance metrics, monitoring protocol adherence, and identifying compliance issues. When deviations or gaps are observed, CPAs support follow-up actions and documentation. CPAs play a key role during study start up activities at trial sites.
| Task | Explanation |
|---|---|
| Support Site Initiation and Setup | CPAs prepare clinical trial sites with required documents, supplies, and equipment to ensure readiness for patient enrolment. |
| Monitor Site Performance and Compliance | CPAs monitor site activities to ensure protocol compliance, track performance metrics, and address any non-compliance issues. |
CPA may not be the right fit if you prefer working independently with minimal coordination, or if frequent follow-ups, timelines, and documentation feel draining. But if you’re organized, proactive, and enjoy keeping complex clinical work on track, this role can be a strong and rewarding starting point in the industry.
4.Data Collection Support & Study Reporting Assistance
Accurate and complete data is the foundation of every clinical trial outcome. CPAs assist in data collection, data entry, and verification, ensuring that trial data is consistent, traceable, and ready for analysis. These activities contribute to ongoing regulatory compliance support.
They also support the preparation of progress reports, safety reports, and final study reports that are reviewed by sponsors and regulatory authorities. While they may not perform statistical analysis, CPAs ensure that data flows correctly from sites to databases and into reports without gaps or inconsistencies.
| Task | Explanation |
|---|---|
| Assist in Data Collection and Entry | CPAs ensure accurate data collection, entry, and verification while maintaining data completeness and consistency. |
| Support Report Preparation | CPAs assist in preparing progress, safety, and final study reports for review by sponsors and regulatory authorities. |
5.Clinical Trial Team Coordination & Information Flow
Clinical trials involve multiple stakeholders project managers, monitors, investigators, sponsors, vendors, and internal teams. CPAs act as communication facilitators, ensuring that information flows clearly and on time.
They organize meetings, circulate updates, track action items, and prepare status reports that help teams stay informed. By supporting internal communication, CPAs reduce confusion, prevent misalignment, and help teams respond quickly to changes or issues.
| Task | Explanation |
|---|---|
| Cross-functional Team Coordination | CPAs coordinate team communication by organizing meetings and sharing project information with stakeholders, sponsors, and site teams. |
| Support Internal Project Team | CPAs support internal project teams with updates, status reports, and clear communication to ensure access to necessary project information. |
6. Protocol, GCP Training Support & Regulatory Awareness
CPAs play a supportive role in training site personnel and internal teams on study protocols, GCP requirements, and trial processes. This ensures that everyone involved understands their responsibilities and follows standardized procedures. This ensures consistent SOP adherence across study teams.
In addition, CPAs are expected to continuously stay updated on regulatory changes, evolving trial methodologies, and industry best practices. This ongoing learning helps them maintain compliance and contribute effectively to trial execution.
| Task | Explanation |
|---|---|
| Assist with Training Site Personnel | CPAs train site staff and project teams on study protocols, GCP standards, and processes to ensure compliance and data accuracy. |
| Stay Updated with Industry Regulations | CPAs stay informed about regulatory changes, trial methodologies, and industry standards to maintain ongoing compliance. |
Clinical Research
Gain in-depth understanding of how clinical trials operate across pharma companies, CROs, and research sites. This program focuses on the practical execution of clinical studies, ethical conduct, documentation, and regulatory compliance across the clinical trial lifecycle.

Duration: 12 months
Skills you’ll build:
Day in the Life of a Clinical Project Associate (CPA)
A Clinical Project Associate begins the day by checking project updates and understanding what needs to be completed to keep the clinical trial on track. They review timelines, follow up on pending tasks, and ensure that study activities are progressing as planned.
As the day continues, the CPA focuses on organizing and updating study documents. Trial files are reviewed for accuracy, new documents are filed correctly, and everything is kept ready for audits and inspections. Documentation control remains a constant responsibility throughout the day.
The CPA then supports coordination between teams by sharing updates, tracking action items, and assisting with meeting follow-ups. Communication with clinical sites and external partners helps ensure that study requirements are met and any issues are documented and addressed.
Before the day ends, the CPA updates trackers and reports, ensuring that all activities are recorded and visible to the project team. By maintaining order, coordination, and compliance, the CPA helps the clinical trial move forward smoothly.
Who Can Become a Clinical Project Associate?
A bachelor’s degree in life sciences, health sciences, pharmacy, nursing, or a related field is required for the role. A master’s degree, such as an MSc in Clinical Research or an MPH, is preferred for more advanced or competitive positions. Certifications or training in clinical research, project management, or regulatory compliance (e.g., GCP certification) are also valuable. Additionally, some prior experience in clinical trial operations, clinical research coordination, data management, or a related field is beneficial.
This role requires strong organizational and communication skills, attention to detail, and basic project management abilities essential for clinical project associate jobs. Knowledge of ICH GCP guidelines with trial tools (CTMS, spreadsheets) are essential for compliance. Adaptability and problem-solving skills are also crucial for managing changing trial demands and operational issues. Familiarity with a clinical trial management system is beneficial.
Conclusion
Clinical Project Associates (CPAs) play a vital role in the smooth execution of clinical trials. They manage key tasks such as documentation, project coordination, site management, and data handling, ensuring that trials stay on track and comply with regulatory standards. CPAs are essential to maintaining trial efficiency, reducing delays, and supporting the timely delivery of new treatments. Their contributions are indispensable for the successful progression of clinical research, making them integral to the overall success of clinical trials.
If you’re interested in pursuing a career in clinical research and becoming a vital part of clinical trials, the PG Diploma in Clinical Research by Clinilaunch providing excellent opportunity to gain the knowledge and skills needed to succeed.
FAQ:
1. Do CPAs work on multiple clinical trials at the same time?
Yes. CPAs often support more than one study simultaneously, depending on trial complexity and organization structure. This helps them gain broader exposure to different trial phases and processes.
2. Is prior industry experience mandatory to become a CPA?
No. Many organizations hire freshers or trained candidates for CPA roles. Relevant training, understanding of clinical trial workflows, and documentation practices are often more important than prior experience.
3. Do CPAs interact directly with regulatory authorities?
CPAs usually do not communicate directly with regulatory authorities. However, they support regulatory submissions and documentation that are reviewed by sponsors or regulatory teams.
4. Can CPAs work in remote or hybrid roles?
Yes. Many CPA roles are office-based or remote, especially in CROs and sponsor companies, as the work primarily involves coordination, documentation, and system-based activities.
5. How is a CPA’s performance usually evaluated?
Performance is commonly assessed based on documentation accuracy, timeline adherence, task follow-up, communication quality, and audit readiness support rather than field-based activities.
6. Is the CPA role more administrative or technical?
The CPA role is operational rather than purely administrative. It requires understanding clinical trial processes, regulatory expectations, and coordination of workflows, not just routine office work.
7. Do CPAs need strong technical or programming skills?
No programming skills are required. Basic proficiency in spreadsheets, document management systems, and clinical trial tools is sufficient for most CPA roles.
8. Can a CPA transition into non-project roles later?
Yes. With experience, CPAs can move into regulatory affairs, trial operations, quality assurance support, or training roles within clinical research organizations.
9. Is the CPA role the same across all companies?
Core responsibilities remain similar, but the exact scope can vary between sponsors, CROs, and research sites depending on organizational structure and trial scale.
10. How long does it usually take to grow from CPA to a senior role?
Typically, professionals can progress to senior or coordination roles within 2–4 years, depending on performance, exposure to multiple studies, and organizational opportunities.