Drug Safety Analyst Career
A Drug Safety Analyst reviews adverse event data. They follow global regulations. They help support patient safety in the drug industry. It is a growing field in pharmacovigilance. It offers strong career growth and global opportunities. Demand is rising due to more drug approvals and safety monitoring needs.
Start Your Drug Safety Career with Industry-Ready Training
The Invisible Shield Between Medicine and Harm
Every medicine used in healthcare carries both benefits and risks. Ensuring that this balance remains safe for patients is the responsibility of pharmacovigilance a critical function within the pharmaceutical industry. At the center of this process is the Drug Safety Analyst. This role monitors, evaluates, and reports adverse drug reactions. These reports support safer healthcare decisions. The Drug Safety Analyst career is crucial and continues to evolve in modern healthcare.
Pharmacovigilance is the science of detecting, assessing, understanding, and preventing adverse effects of medicines throughout their lifecycle from clinical trials to post-market use. It plays a vital role in maintaining regulatory compliance and protecting patient safety, making it an essential pillar of modern healthcare systems.
The importance of drug safety has increased with the complexity of modern therapeutics, where drugs tested on limited populations are later used by millions with diverse conditions. According to the World Health Organization, adverse drug reactions account for nearly 5–7% of hospital admissions globally. At the same time, rising drug approvals over 46.5 annually by the U.S. Food and Drug Administration and stricter regulations from agencies like the European Medicines Agency have made continuous safety monitoring essential. In this landscape, Drug Safety Analysts play a critical role in analyzing adverse events, ensuring compliance, and supporting safer healthcare decisions through data-driven insights.
To build expertise in this evolving domain, specialized training programs such as the Advanced Diploma in AI Integration in Drug Safety & Compliance offered by CliniLaunch Research Institute are designed to equip learners with practical pharmacovigilance knowledge, regulatory understanding, and AI-driven skills required for modern drug safety roles.
What is Pharmacovigilance?
No drug is born perfect. Even after years of clinical trials, a medicine enters the real-world carrying unknowns rare side effects, drug interactions, population-specific risks that only surface at scale. Pharmacovigilance (PV) is the scientific discipline built to catch exactly that.
Formally defined by the WHO, Pharmacovigilance is the science and activities relating to the detection, assessment, understanding and prevention of adverse effects or any other medicine-related problems. It operates across the entire drug lifecycle from Phase I trials to decades of post-market surveillance.
Why it exists:
- Patient Safety : Ensures that benefits of a drug consistently outweigh its risks in real-world use
- Regulatory Compliance : FDA, EMA, and CDSCO mandate structured safety reporting; non-compliance means market withdrawal, hefty fines, or criminal liability
- Post-Market Risk Management Approximately 1 in 3 drugs approved receives a post-market safety label change Market Data Forecast, proving that trial data alone is never the full picture
Where PV fits in the ecosystem:
Think of Pharma as a value chain R&D discovers, clinical trials validate, regulatory affairs approves, and commercial teams sell. Pharmacovigilance is the continuous safety net running parallel to all of it from first-in-human studies to the drug’s last day on the market.
It connects directly with:
- Regulatory Affairs — for submission of safety reports (ICSRs, PSURs)
- Clinical Operations — for trial-phase adverse event monitoring
- Medical Affairs — for risk communication to healthcare professionals
With over 3 million individual case safety reports (ICSRs) submitted to global databases annually Fortune Business Insights, PV is no longer a compliance checkbox, it is a data-intensive, technology-driven discipline that sits at the intersection of science, law, and public health.
Pharmacovigilance Process
The pharmacovigilance process follows a structured workflow to ensure continuous monitoring of drug safety in real-world use.
- Case Intake – Adverse event reports are received from healthcare professionals, patients, or clinical trials.
- Case Processing – Data is entered, validated, and structured into standardized safety reports (ICSRs).
- Medical Review – Clinical experts assess the case for seriousness, expectedness, and medical relevance.
- Signal Detection – Patterns are analyzed across multiple cases to identify potential safety risks.
- Regulatory Reporting – Verified safety data is submitted to global authorities such as the U.S. Food and Drug Administration and the European Medicines Agency within defined timelines.
This workflow is where Drug Safety Analysts play a key operational and analytical role.
Who is a Drug Safety Analyst & Where Do They Fit?
A Drug Safety Analyst (DSA) is a trained professional. They collect, process, evaluate, and report adverse drug event data. They follow global regulatory standards. They are the first line of scientific judgment in determining whether a drug is behaving safely in the real world.
In simple terms: if pharmacovigilance is the system, the Drug Safety Analyst is the engine running it.
Where They Work:
- Pharmaceutical & Biotech Companies — In-house PV departments managing their own drug portfolios (Pfizer, Sun Pharma, Dr. Reddy’s)
- Contract Research Organizations (CROs) — Handling PV outsourcing for multiple pharma clients simultaneously (ICON, Covance, Syneos Health)
- Knowledge Process Outsourcing firms (KPOs) — India’s fastest-growing DSA employment segment; companies like Accenture Life Sciences, Cognizant, and Wipro BPS run large PV delivery centers
- Regulatory Agencies — FDA, EMA, and CDSCO employ safety reviewers who evaluate submissions from pharma companies
- Hospitals & Academic Centers Pharmacovigilance units monitoring ADRs from clinical practice and investigator-initiated trials
How They Connect to the PV Workflow:
A DSA doesn’t just touch one step they operate across the entire pipeline:
PV Workflow: Role of a Drug Safety Analyst
| PV Stage | DSA’s Role |
|---|---|
| Case Intake | Triaging and logging incoming adverse event reports |
| Case Processing | MedDRA coding, ICSR documentation, database entry |
| Medical Review | Supporting narrative writing and causality assessment |
| Signal Detection | Running aggregate data queries, flagging patterns |
| Regulatory Reporting | Preparing and submitting ICSRs, PSURs within deadlines |
India has become a global pharmacovigilance powerhouse handling critical drug safety operations for companies worldwide making it the second-largest pharmacovigilance delivery hub globally after the United States, driven by its large English-speaking, scientifically trained workforce.
Roles of a Drug Safety Analyst
Drug Safety Analysts are responsible for managing and evaluating adverse event data throughout the pharmacovigilance process, ensuring accuracy, compliance, and timely reporting. Their role combines data handling, clinical understanding, and regulatory responsibility to maintain drug safety in real-world settings.
Gain Hands-On Skills in Pharmacovigilance & Drug Safety

1. Case Handling & Processing Responsibilities
Case Intake & Triage
- Receiving adverse event reports from multiple sources and prioritizing them based on seriousness and reporting timelines.
- Example: A serious adverse event (SAE), including life-threatening events, is prioritized for immediate reporting (typically within 24 hours as per protocol or SOP requirements) and expedited regulatory submission in accordance with ICH and FDA guidelines.
Case Data Entry & Medical Coding (MedDRA, WHO-DRL)
- Entering case details into safety databases and coding adverse events and drugs using standardized dictionaries like MedDRA and WHO-DRL.
- Example: A reported “severe headache” is accurately coded to the appropriate MedDRA term to ensure consistency in global safety databases.
Case Processing (ICSR Handling)
- Structuring and validating Individual Case Safety Reports (ICSRs) to ensure completeness and regulatory compliance.
- Example: A case with incomplete patient information is flagged and processed only after verifying mandatory fields required for regulatory submission.
2. Clinical Evaluation & Documentation
Narrative Writing & Medical Summarization
- Creating clear and concise narratives summarizing patient history, adverse events, and outcomes.
- Example: A multi-drug case is summarized into a structured narrative highlighting timelines, suspected drug, and clinical outcome for medical review.
Case Assessment & Basic Causality Evaluation
- Reviewing clinical data to support initial assessment of the relationship between the drug and the adverse event.
- Example: An adverse reaction occurring shortly after drug administration is assessed for temporal association and flagged for further review.
3. Follow-up & Coordination
Follow-ups & Query Management
- Identifying missing or unclear data and coordinating with reporters (physicians or sites) to obtain additional information.
- Example: A case lacking dosage details is followed up with the reporting physician to ensure accurate evaluation and reporting.
4. Quality & Compliance Responsibilities
Quality Check & Accuracy Review
- Performing detailed reviews to ensure data accuracy, consistency, and completeness before submission.
- Example: A discrepancy between patient age in two sections of the report is identified and corrected during quality review.
Compliance with Global Regulatory Guidelines
- Ensuring all cases meet international regulatory standards. These standards include those from the U.S. Food and Drug Administration and the European Medicines Agency.
- Example: A serious unexpected adverse event is processed and documented as per ICH guidelines to meet global compliance requirements.
5. Regulatory & Timeline Management
Regulatory Reporting
- Submitting validated safety reports to regulatory authorities within defined formats and requirements.
- Example: A validated ICSR is submitted electronically to regulatory databases like EudraVigilance within the required framework.
Adherence to Strict Reporting Timelines
- Managing timelines for reporting serious and non-serious cases to avoid compliance risks.
- Example: A fatal adverse event is reported within the mandated 7-day timeline to meet regulatory obligations.
Skills Required for Drug Safety Analyst
To succeed as a Drug Safety Analyst, professionals need clinical knowledge, technical expertise, and analytical skills. These skills help them manage and evaluate drug safety data.
Core Skills
- Medical terminology – Understanding clinical terms, conditions, and drug-related concepts for accurate case interpretation.
- Attention to detail – Ensuring precision while handling sensitive safety data and identifying inconsistencies.
- Documentation & narrative writing – Structuring clear and concise case narratives for medical and regulatory review.
- Critical thinking – Analyzing case information logically to support safety evaluation and decision-making.
Technical Skills
- Case processing (ICSR handling) – Managing and structuring adverse event reports as per pharmacovigilance standards.
- MedDRA coding – Classifying adverse events using standardized medical terminology for global consistency.
- Safety databases (Argus / Veeva / ArisG) – Working with industry tools to record, track, and manage safety data.
- Basic regulatory knowledge (ICH, U.S. Food and Drug Administration, European Medicines Agency) – Understanding global guidelines for compliant reporting and documentation.
Advanced Skills (Modern & Industry-Relevant)
- Signal detection & risk evaluation – Identifying patterns in safety data to detect potential drug risks.
- AI & automation in pharmacovigilance (NLP-based workflows) – Leveraging AI tools to streamline case processing and data extraction.
- Aggregate reporting (PSUR, PBRER) – Contributing to periodic safety reports that evaluate overall drug benefit risk profiles.
- Global regulatory intelligence – Staying updated with evolving international drug safety regulations and requirements.
- Safety data analytics (Excel / Power BI) – Analyzing large datasets to derive insights for safety monitoring and decision-making.
These skills reflect the evolving nature of pharmacovigilance, where professionals are expected to move beyond processing into analysis, interpretation, and technology-driven workflows.
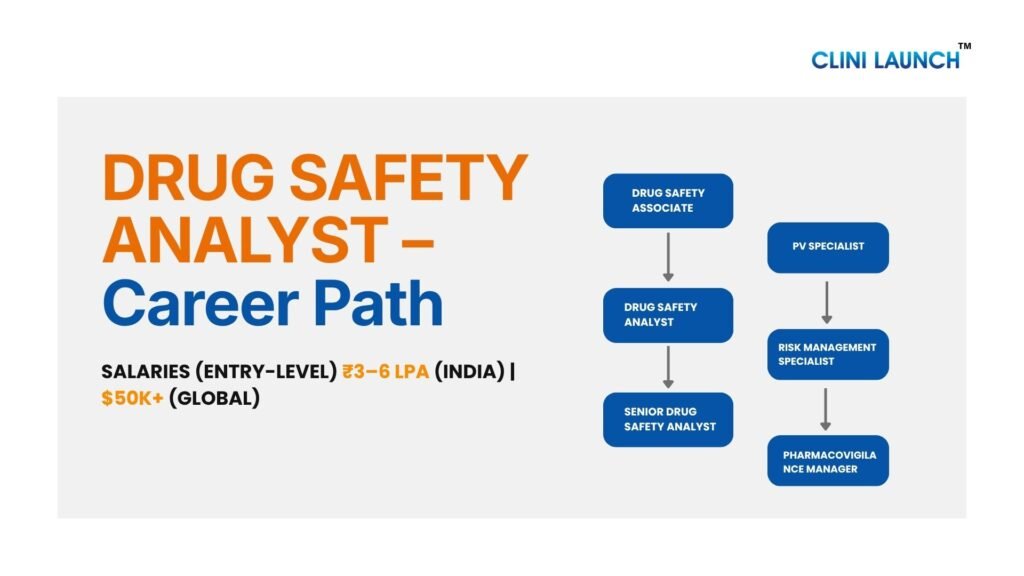
Career Path in Pharmacovigilance
A career in drug safety progresses from operational roles to analytical and leadership positions within pharmacovigilance. Professionals often start as Drug Safety Associates, focusing on case processing. They can advance to Senior Drug Safety Analysts, handling complex cases and quality reviews. With experience, they move into roles such as Pharmacovigilance Specialists or Team Leads. Later, they can advance to roles like Risk Management Specialists or Pharmacovigilance Managers. In these roles, they support safety strategy and help with regulatory decisions.
Drug Safety Analyst Salary in India Vs Global:
Drug Safety Analyst Salary in India vs Global
| Job Role | Experience Level | India (LPA) | Global Salary (Approx) |
|---|---|---|---|
| Drug Safety Associate | Entry Level (0–2 Years) | 3 – 5 LPA | $50,000 – $80,000 / year |
| Drug Safety Analyst | 0–4 Years | 4 – 6 LPA | $60,000 – $75,000 / year |
| Senior Drug Safety Analyst | 3–8 Years | 5 – 8 LPA | $70,000 – $100,000 / year |
Key Insight
- Entry-level salaries in India typically range from ₹3–6 LPA, depending on skills and company
- Globally, pharmacovigilance professionals earn significantly higher, with entry roles starting around $45,000–$65,000+ annually
- With experience, global salaries can exceed $100,000+, especially in the US and Europe
This clearly shows that drug safety is not just a stable career—it is a globally scalable profession, offering strong growth in both responsibility and compensation as you gain experience.
Lateral Moves Available
A Drug Safety Analyst’s skill set is highly transferable — opening doors beyond the core pharmacovigilance (PV) track:
- Regulatory Affairs — leveraging regulatory knowledge for drug approvals
- Medical Writing — using narrative and documentation expertise
- Clinical Research / CRA — transitioning into trial-phase safety monitoring
- PV Consulting — independent advisory roles for pharma and biotech startups
AI Integration in Drug Safety & Compliance
Learn how artificial intelligence is applied in pharmacovigilance, drug safety monitoring, and regulatory compliance. This program focuses on real-world tasks like adverse event processing, signal detection, and AI-driven safety data analysis. It is used in global healthcare settings.

Duration: 6 months
Learn at your own pace
Skills you’ll build:
Job Opportunities & Industry Demand
The demand for Drug Safety Analysts is not cyclical it is structurally driven. Every new drug approval adds to the safety work that must be done.Each expanding clinical pipeline adds more safety work.Every tighter regulatory mandate also increases the safety work needed. And that volume is only growing.
🇮🇳 India The World’s Fastest-Growing PV Hub
India is no longer just a cost-efficient outsourcing destination. It has become a strategic delivery center for global pharmacovigilance operations powered by its large English-speaking, scientifically trained graduate pool and a maturing pharma ecosystem.
Key Hiring Cities and Their PV Ecosystem
| City | PV Ecosystem |
|---|---|
| Bengaluru | KPO-led delivery; Indegene, Accenture Life Sciences, TCS |
| Hyderabad | CRO-heavy; PAREXEL, Syneos Health, Makrocare, Vigimedsafe |
| Pune | Mixed pharma + CRO; Cognizant, Syneos, mid-size Indian pharma |
| Mumbai | Corporate pharma HQs; Sun Pharma, Cipla, Dr. Reddy’s |
KPO-led delivery; Indegene, Accenture Life Sciences, TCS
CRO-heavy; PAREXEL, Syneos Health, Makrocare, Vigimedsafe
Mixed pharma + CRO; Cognizant, Syneos, mid-size Indian pharma
Corporate pharma HQs; Sun Pharma, Cipla, Dr. Reddy’s
A Global PV Delivery Powerhouse
India has evolved from a cost-effective outsourcing destination to a strategic pharmacovigilance hub for global pharma.
Multinational pharmaceutical companies have established dedicated PV centers in India to manage global drug safety operations— meaning the work being done from Bengaluru and Hyderabad today is directly feeding regulatory submissions to the FDA and EMA. Global CROs often hold walk-in hiring drives in Hyderabad, Bengaluru, and Pune Market Data Forecast.These cities stay busy hiring fresh graduates all year.
India’s PV salaries are rising at 12–15% annually— one of the fastest growth rates globally — reflecting how rapidly the country is moving from volume delivery to higher-complexity PV work like signal detection and aggregate reporting.
Global Trends
Globally, demand for pharmacovigilance professionals is rising. This is because more drugs are getting approved.Agencies like the U.S. Food and Drug Administration and the European Medicines Agency approve these drugs. This leads to more post-marketing safety data. At the same time, stricter rules and ongoing safety monitoring are expanding the scope of drug safety roles. A significant portion of pharmacovigilance activities is also being outsourced to countries like India, further strengthening its position as a global drug safety hub.
This growing demand highlights pharmacovigilance as a stable and globally relevant career path, with expanding opportunities across both Indian and international markets.
Future Outlook
Pharmacovigilance is changing due to new technology, more data, and strict global rules. This shift is shaping a more active role for Drug Safety Analysts.
AI in Pharmacovigilance
AI and NLP are improving efficiency in case processing, data extraction, and signal detection.
- Example: AI tools can scan thousands of patient reports or literature sources to automatically identify potential adverse events for further review.
Automation vs Human Expertise
Automation handles repetitive tasks, but clinical judgment and decision-making still rely on human expertise.
- Example: While software auto-populates case data, a Drug Safety Analyst evaluates whether the drug caused the adverse reaction.
Increasing Regulatory Complexity
Regulations from agencies like the U.S. Food and Drug Administration and the European Medicines Agency keep changing. This increases the need for skilled professionals.
- Example: A change in reporting timelines requires analysts to update workflows to ensure serious cases are submitted within new regulatory deadlines.
Long-Term Stability of the Role
With growing drug approvals and ongoing safety monitoring needs, drug safety remains a stable and future-relevant career.
- Example: Every newly approved drug requires continuous safety monitoring, creating consistent demand for pharmacovigilance professionals.
Become a Job-Ready Drug Safety Analyst in 6 Months
Conclusion
A career as a Drug Safety Analyst is ideal for people with a life sciences background. It fits those who are detail-oriented and analytical. It also suits those interested in healthcare, data, and regulatory science. It particularly suits those who want to contribute to patient safety while building a stable and globally relevant career.
Pharmacovigilance is a strong healthcare career option. It is not limited to one phase of drug development. It spans a medicine’s full lifecycle. With more drug approvals, stricter rules, and greater use of real-world data, demand for drug safety experts is rising. This trend is growing in India and worldwide. In a world driven by innovation, Drug Safety Analysts ensure that progress never comes at the cost of patient safety.
For freshers and life science graduates, this field offers a clear entry into healthcare. It has structured roles and clear career paths. It also offers chances to move into advanced areas. These include regulatory affairs, clinical research, and risk management.
To build a career in this evolving domain, gaining the right skills and practical exposure is essential. Programs like the Advanced Diploma in AI Integration in Drug Safety & Compliance offered by CliniLaunch Research Institute are designed to equip learners with industry-relevant knowledge, regulatory understanding, and hands-on experience in modern pharmacovigilance practices.
Frequently Asked Questions
It directly contributes to patient safety by ensuring that medicines are monitored and evaluated throughout their lifecycle.
Life science backgrounds such as Pharmacy, Biotechnology, and Microbiology provide a strong foundation.
By analyzing adverse event data, they help identify risks early and support safer treatment decisions globally.
Professionals use safety databases, medical coding systems, and AI-driven tools for data analysis.
Yes, it is a globally standardized field with opportunities across pharmaceutical companies, CROs, and regulatory environments.
It works with clinical research, regulatory affairs, and medical teams to ensure drug safety across all stages.
The field offers structured progression from entry-level roles to specialized and leadership positions.
AI, automation, and data analytics are enhancing efficiency and expanding the analytical scope of the role.
Pharmaceutical companies, CROs, healthcare organizations, and consulting firms actively recruit for these roles.
With increasing drug development and stricter safety regulations, the demand for continuous monitoring ensures long-term career relevance.