| Regulatory Affairs Associate Role in Clinical Trials |
|---|
| A Regulatory Affairs Associate supports clinical trials by preparing and maintaining regulatory documentation, coordinating regulatory and ethics submissions, tracking approvals and amendments, and ensuring ongoing compliance from study start-up through close-out. |
Clinical research and product development don’t move forward just because science looks promising. Every study, drug, or medical product must pass defined regulatory checks before it can start, continue, or close. Someone has to make sure those rules are followed consistently and on time. That responsibility sits with regulatory teams, and at the entry level, with the Regulatory Affairs Associate.
A Regulatory Affairs Associate (RAA) supports the regulatory process by managing documentation, tracking approvals, coordinating inputs across teams, and ensuring compliance with approved requirements.
Regulatory Affairs Associate positions exist across pharmaceutical companies, biotechnology firms, clinical research organizations, and medical device companies. These professionals operate at the intersection of science, operations, and regulation, supporting studies and products throughout their lifecycle.
This blog focuses strictly on the real-world responsibilities of a Regulatory Affairs Associate. You’ll learn where the role fits in clinical research, the core responsibility areas handled at the associate level, what the role does not include, and the skills used on the job. The goal is clarity, so you can judge early whether this role genuinely fits your career direction.
What does a Regulatory Affairs Associate Do?
A regulatory affairs associate operates at the execution layer of clinical research and product development. The role sits between scientific teams and regulatory authorities, ensuring that approved plans are followed exactly as required under applicable clinical trial regulations.
In practical terms, an RAA is involved across the entire clinical trial lifecycle:
- Before a study starts: supporting regulatory and ethics submissions, tracking approvals, and confirming readiness to begin
- While the study is ongoing: managing amendments, maintaining compliance, and keeping regulatory documentation inspection-ready
- After the study ends: supporting close-out notifications, final documentation, and post-study regulatory readiness
RAAs execute regulatory tasks under supervision, ensuring nothing moves forward without the required approvals in place.
From a career standpoint, this is not a niche or academic role. Regulatory Affairs Associate positions exist across multiple parts of the healthcare industry:
- Pharmaceutical companies delivering regulatory compliance in pharmaceuticals such as Sun Pharma and Dr. Reddy’s
- Biotechnology company’s firms navigating complex drug regulatory affairs like Biocon
- Clinical Research Organizations (CROs) offering end-to-end regulatory affairs services including IQVIA and Syneos Health
- Medical device companies supporting global approvals such as Medtronic
Across all these settings, the role stays fundamentally the same: support regulatory compliance, so studies and products can move forward without delays, findings, or legal risk.
This positioning is important for beginners to understand early. A Regulatory Affairs Associate is not a “background” role. It is an operational control role, where small errors can have large downstream consequences.
Why Can’t Clinical Research Move Forward Without Regulatory Teams?
Clinical research isn’t just about science and discovery it’s about doing things the right way. Every clinical trial must meet strict regulatory rules and safety standards before it can begin or continue. Without proper oversight on regulations, approvals, and compliance, trials can face delays, increased costs, or even be halted entirely.
For example, research analyzing global randomized clinical trials has shown that regulatory and administrative processes including ethics committee approvals and country-specific submission requirements are among the primary drivers of trial start-up delays, significantly affecting timelines and study progression. This demonstrates how critical regulatory oversight is in ensuring that trials move forward efficiently and compliantly.
This rigorous regulatory environment exists to protect participants, maintain data integrity, and make sure that only safe and effective treatments progress — a responsibility that cannot be fulfilled without dedicated regulatory teams guiding the way.
How Do Regulatory Affairs Associates Support Regulatory Submissions?
Once the importance of regulatory oversight in clinical research is clear, the next question is how this support is delivered in practice. Regulatory Affairs Associates play a critical role by getting involved early in the regulatory submission process, well before applications are filed with authorities. They work closely with clinical, quality, and manufacturing teams to ensure that required data, documents, and timelines align with regulatory expectations from the start.
By supporting submission planning, reviewing documentation for accuracy and consistency, and tracking regulatory requirements, they help reduce the risk of last-minute gaps that can delay approvals. This early involvement is essential because regulatory submissions are built progressively, and issues identified late in the process are often costly and difficult to correct. Through this structured support, Regulatory Affairs Associates help submissions move forward smoothly and with greater confidence.
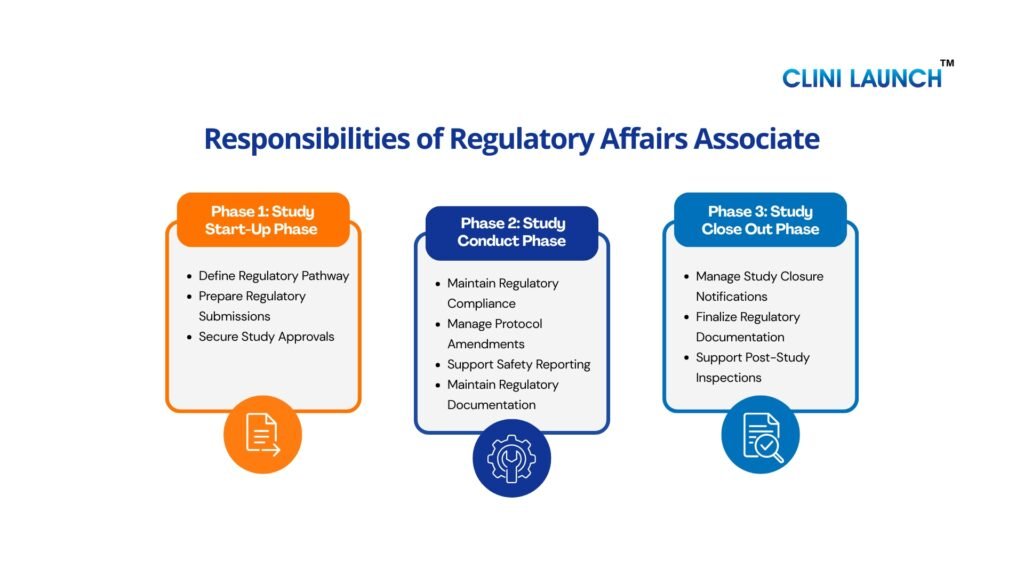
Roles and Responsibilities of a Regulatory Affairs Associate Across Clinical Trial Phases
1. Study Start-Up Phase
The Study Start-Up Phase determines whether a clinical study can begin legally and ethically. Regulatory Affairs Associates ensure study design, documentation, and submission plans meet regulatory requirements. Gaps at this stage can delay approvals or prevent site activation.
1. Defining the Study’s Regulatory Pathway
At the beginning of a study, Regulatory Affairs Associates help interpret regulatory requirements based on the country of conduct, study phase, and study type. They ensure that the study follows the correct regulatory pathway and meets ethical standards before submissions begin.
- Identify applicable regulations and guidance documents
- Ensure alignment with local and global regulatory expectations
- Support teams in understanding regulatory constraints early
2. Preparing the Study for Regulatory and Ethics Approval
Regulatory Affairs Associates play a key role in preparing and coordinating initial submissions to regulatory authorities and Ethics Committees / IRBs. Their responsibility is to ensure that submission packages are complete, accurate, and compliant before they are filed.
- Assist in compiling submission-ready documents
- Coordinate inputs from clinical and other internal teams
- Ensure submission formats and content meet authority requirements
3. Securing Approvals to Activate the Study
Once submissions are made, Regulatory Affairs Associates track approval of progress and support responses to authority or ethics queries. They ensure that all required approvals are obtained before studying activation or subject enrollment.
- Monitor approval timelines
- Coordinate responses to clarification requests
- Confirm formal approvals before trial initiation
Example scenario: Study Start-Up: Regulatory Support in Action
Problem:
A multi-country clinical study faced potential delays due to differences in regulatory and ethics requirements across regions.
Action:
The Regulatory Affairs Associate first focused on defining the study’s regulatory pathway by identifying country-specific regulatory expectations and appropriate submission routes. Next, they supported preparing the study for regulatory and ethics approval by coordinating submission-ready documents and aligning inputs from clinical teams. After submissions were filed, the Associate supported securing approvals to activate the study by tracking review timelines and coordinating responses to authority and ethics queries.
Outcome:
All required approvals were obtained on time, allowing smooth site activation and preventing delays during study start-up.
2. Study Conduct Phase
The Study Conduct Phase begins once the clinical study is activated and continues until the last subject visit is completed. During this phase, Regulatory Affairs Associates ensure that the study is conducted strictly according to approved protocols, regulatory requirements, and ethics approvals. Any unapproved changes or compliance gaps during this stage can lead to regulatory findings, delays, or study suspension.
1. Maintaining Ongoing Regulatory Compliance
During study conduct, Regulatory Affairs Associates support continuous compliance by ensuring that trial activities remain aligned with approved regulatory and ethics conditions. They help monitor adherence to protocols and regulatory commitments throughout the study lifecycle.
- Ensure study activities follow approved protocols and submissions
- Monitor compliance with regulatory and ethics conditions
- Support teams in understanding regulatory obligations during conduct
2. Managing Protocol Amendments and Study Changes
Clinical studies often require changes due to operational or safety reasons. Regulatory Affairs Associates support the preparation, review, and submission of protocol amendments and related documents to ensure changes are approved before implementation.
- Support preparation of protocol amendments and updated documents
- Coordinate amendment submissions to authorities and Ethics Committees / IRBs
- Ensure approvals are obtained before changes are implemented
3.Supporting Safety and Regulatory Reporting
Regulatory Affairs Associates assist with ongoing regulatory reporting obligations during the study, particularly related to safety updates and regulatory notifications. Their role helps ensure timely and compliant communication with authorities.
- Support regulatory submissions related to safety updates
- Ensure reporting timelines are met
- Coordinate inputs from safety and clinical teams
- Maintaining Regulatory Documentation and Records
Accurate and up-to-date regulatory documentation is critical during study conduct. Regulatory Affairs Associates help ensure that regulatory files remain current, traceable, and inspection-ready throughout the study.
- Maintain regulatory records and submission histories
- Ensure document version control and traceability
- Support audit and inspection readiness
Example Scenario: Study Conduct – Regulatory Oversight in Practice
Problem:
During an ongoing clinical study, operational updates and safety-related changes created a risk of non-compliance if implemented without proper regulatory approval.
Action:
The Regulatory Affairs Associate supported maintaining ongoing regulatory compliance by ensuring study activities continue to align with approved protocols and regulatory conditions. When changes were required, the Associate supported managing protocol amendments by coordinating updated documents and submissions to regulatory authorities and Ethics Committees. In parallel, they supported safety and regulatory reporting by tracking reporting timelines and coordinating inputs from clinical and safety teams, while ensuring regulatory documentation remained current and audit-ready throughout the study.
Outcome:
All study changes and reports were managed in compliance with regulatory requirements, allowing the study to continue without findings, interruptions, or regulatory delays
C. Study Close-Out Phase
The Study Close-Out Phase begins once the last subject visit is completed and continues until all regulatory and ethics obligations are formally closed. During this phase, Regulatory Affairs Associates ensure that the study is properly concluded in compliance with regulatory requirements and that all records are finalized, complete, and inspection ready. Incomplete close-out activities can lead to regulatory findings even after a study has ended.
- Managing End-of-Study Regulatory and Ethics Notifications
At study completion, Regulatory Affairs Associates support required notifications to regulatory authorities and Ethics Committees / IRBs to formally communicate study closure.
- Support preparation of end-of-study notifications
- Coordinate submissions to regulatory authorities and Ethics Committees / IRBs
- Ensure notifications are submitted within required timelines
- Finalizing Regulatory Documentation and Records
Regulatory documentation must be complete and accurate at the time of study close-out. Regulatory Affairs Associates verify that all regulatory records reflect the final status of the study.
- Review regulatory files for completeness and consistency
- Ensure all approvals, amendments, and correspondence are finalized
- Support proper archiving of regulatory documentation
- Supporting Regulatory Inspections After Study Completion
Even after a study is closed, regulatory inspections may still occur. Regulatory Affairs Associates help ensure continued inspection of readiness post close-out.
- Ensure regulatory records remain accessible and traceable
- Support document retrieval during inspections or audits
- Assist with responses to post-study regulatory queries
Example Scenario: Study Close-Out – Regulatory Completion in Action
Problem:
As a clinical study reached completion, there was a risk of regulatory non-compliance if end-of-study notifications, documentation finalization, and regulatory records were not handled correctly or on time.
Action:
The Regulatory Affairs Associate supported regulatory close-out activities by coordinating end-of-study notifications to regulatory authorities and Ethics Committees. They ensured regulatory documentation was complete and accurate by verifying that all approvals, amendments, and correspondence were properly finalized and archived. The Associate also supported inspection of readiness after study completion by ensuring regulatory records remained accessible and audit-ready, even after the study formally ended.
Outcome:
All regulatory close-out requirements were completed within timelines; documentation was properly archived, and the study remained inspection-ready, ensuring long-term regulatory compliance after study completion.
Skills Required for a Regulatory Affairs Associate
Regulatory & Technical Skills
These skills form the foundation of a Regulatory Affairs Associate’s role. They ensure that clinical studies and regulatory submissions align with applicable laws, guidelines, and ethical standards. Strong regulatory knowledge helps prevent compliance gaps and approval delays from the outset.
- Understanding of Clinical Trial Regulations and Guidelines
Ensures studies follow approved legal and ethical frameworks. Helps identify what is permitted at each stage and prevent compliance gaps. Reduces approval delays and regulatory risk.
- Knowledge of Regulatory and Ethics Submission Processes
Supports accurate preparation and filing of regulatory and ethics submissions. Ensure completeness and alignment with authority expectations. Enables timely approvals and smooth study progression.
- Familiarity with Protocols, ICFs, Amendments, and Regulatory Documentation
Ensure consistency across core clinical trial documents. Helps identify gaps or discrepancies early. Maintains regulatory accuracy throughout the study lifecycle.
Operational Skills
Operational skills enable Regulatory Affairs Associates to translate regulatory requirements into day-to-day execution. They help manage coordination across teams, track regulatory timelines, and ensure regulatory activities move forward smoothly. These skills are critical for keeping studies on schedule without compliance risks.
- Coordination with Clinical, Quality, and Safety Teams
Enables a timely collection of regulatory inputs from multiple teams. Ensure alignment of data and documentation. Prevents delays caused by missing or inconsistent information.
- Tracking Approvals, Timelines, and Regulatory Queries
Ensures regulatory milestones and responses are not missed. Supports timely follow-up with authorities and teams. Keep studying on schedule.
- Maintaining Audit-Ready Regulatory Records
Keeps regulatory documentation complete and traceable. Supports inspections during and after the study. Demonstrates ongoing compliance.
Professional Skills
Professional skills determine how effectively a Regulatory Affairs Associate performs under regulatory pressure. Accuracy, communication, and time management are essential in a role where small errors can have significant regulatory impact. These skills support consistent performance in high-accountability environments.
- Attention to Detail
Prevents documentation errors and compliance issues. Ensure accuracy across submissions and records. Critical for avoiding regulatory findings.
- Clear Written Communication
Ensures regulatory information is clearly understood and applied. Supports accurate documentation and responses. Enables effective cross-team coordination.
- Working Under Regulatory Timelines and Pressure
Helps manage multiple deadlines without compromising compliance. Supports consistent performance in high-risk regulatory environments. Essential for maintaining study momentum.
Clinical Research
Develop industry-ready clinical research skills used across pharmaceutical companies, CROs, and healthcare organizations. Learn how clinical trials are designed, conducted, monitored, and regulated, while gaining hands-on exposure to real-world clinical research workflows and compliance standards.

Duration: 6 months
Learn at your own pace
Skills you’ll build:
Regulatory Affairs Associate Salary by Organization Type
| Organization Type | Typical Experience Range | Salary Range (INR) | Approx. Salary Range (USD) |
|---|---|---|---|
| CROs | 0–4 years | ₹3.5 – ₹8 LPA | $4,200 – $9,600 |
| Pharmaceutical Companies | 1–5 years | ₹4 – ₹9 LPA | $4,800 – $10,800 |
| Biotechnology Firms | 2–6 years | ₹5 – ₹10 LPA | $6,000 – $12,000 |
| Specialized / Global Regulatory Roles | 5+ years | ₹9 – ₹12 LPA+ | $10,800 – $14,500+ |
Ready to Build a Career in Regulatory Affairs?
The role of a Regulatory Affairs Associate is central to how clinical research moves forward ensuring studies are compliant, approvals are secured on time, and regulatory risks are managed throughout the study lifecycle. From study start to close-out, regulatory professionals play a key role in maintaining ethical standards, data integrity, and regulatory credibility.
For those looking to build a career in regulatory affairs or clinical research, gaining structured, industry-aligned training is essential. Programs like Advanced Diploma in Clinical Research are designed to bridge the gap between academic learning and real-world regulatory practice. With a strong focus on practical workflows, documentation, and compliance, this program can help you prepare roles such as Regulatory Affairs Associate and take a confident step into the clinical research industry.
FAQ’S
- Who is a regulatory affairs associate?
A Regulatory Affairs Associate ensures that clinical trials and healthcare products comply with regulatory and ethical requirements. They support approvals, submissions, and ongoing compliance.
- What is the role of a regulatory affairs associate?
The role involves supporting regulatory submissions, tracking approvals, managing changes, and maintaining compliance across the clinical trial lifecycle.
- What skills are needed in regulatory affairs?
Key skills include regulatory knowledge, documentation accuracy, coordination across teams, and the ability to work under strict compliance timelines.
- What do regulatory affairs associates do?
They support regulatory and ethics submissions, manage approvals and amendments, and maintain audit-ready regulatory documentation.
- What is the primary role of regulatory affairs?
The primary role is to ensure products and studies meet regulatory requirements so they can be approved, conducted, and marketed legally and safely.
- What are regulatory responsibilities?
Regulatory responsibilities include ensuring compliance, managing approvals, maintaining documentation, and supporting inspections and audits.
- What are regulatory affairs in pharma jobs?
These roles focus on ensuring drugs meet regulatory requirements from development through approval and post-market compliance.
- What are the two important functions of the regulatory affairs department?
Ensuring regulatory compliance and securing timely approvals from regulatory authorities.
- What is the difference between regulatory affairs and compliance?
Regulatory affairs focus on approvals and interactions with regulators, while compliance ensures ongoing adherence to approved regulations and standards.
- What is the purpose of regulatory agencies?
Regulatory agencies protect public health by enforcing safety, quality, and ethical standards across clinical research and healthcare products.