A Safety Reporting Coordinator in clinical trials is responsible for collecting, tracking, and reporting adverse events (AEs), serious adverse events (SAEs), and SUSARs within regulatory timelines. Their role includes ensuring patient safety, maintaining compliance with global regulations, and communicating safety data to sponsors, investigators, and regulatory authorities.
Imagine a clinical trial where a participant reports chest pain… but the information reaches the safety team late. In clinical research, timing and accuracy can directly impact patient safety. Thousands of adverse events are reported during clinical trials every year, and each one must be tracked, verified, and reported within strict regulatory timelines. Even a small delay or missing detail can raise serious concerns during audits and inspections.
The importance of accurate safety reporting becomes clear when we consider the global burden of patient safety issues. According to the World Health Organization (WHO), approximately 1 in every 10 patients is affected in healthcare settings, and more than 50% of that harm is related to medication, many of which could be preventable with proper safety monitoring and reporting. Safety reporting is closely connected with clinical trial data management and contributes to the final clinical study report, ensuring accurate documentation of safety outcomes.
To prevent similar gaps in clinical research, the role of a Drug Safety Reporting Coordinator exists. This position ensures that safety information from clinical trials is collected, followed up systematically, and reported on time, forming a critical part of clinical trials safety reporting that allows potential risks to be identified early while meeting regulatory expectations.
Who is a Safety Reporting Coordinator?
A Safety Reporting Coordinator, often referred to in some organizations as a clinical safety coordinator, is a professional who works within pharmacovigilance or drug safety teams, supporting the accurate and timely reporting of safety information generated during clinical trials. Their primary responsibility is not to medically assess patient safety, but to coordinate and manage the safety reporting process from start to finish.
In day-to-day work, a Safety Reporting Coordinator acts as a central link between multiple stakeholders, including clinical trial sites, sponsors, contract research organizations (CROs), and regulatory authorities. Because safety data flows from different sources, this role ensures that information is collected consistently, followed appropriately, and communicated to the concerned parties without delay.
The role mainly focuses on activities related to Individual Case Safety Reports (ICSRs) and SAE reporting in clinical trials, with close attention to regulatory compliance timelines. Safety Reporting Coordinators track when cases are received, monitor submission deadlines, and follow up on missing or incomplete information to ensure reports meet regulatory requirements.
With experience, professionals can also progress into roles such as clinical safety officers within pharmacovigilance teams.
Trusted by 2,500+ Learners
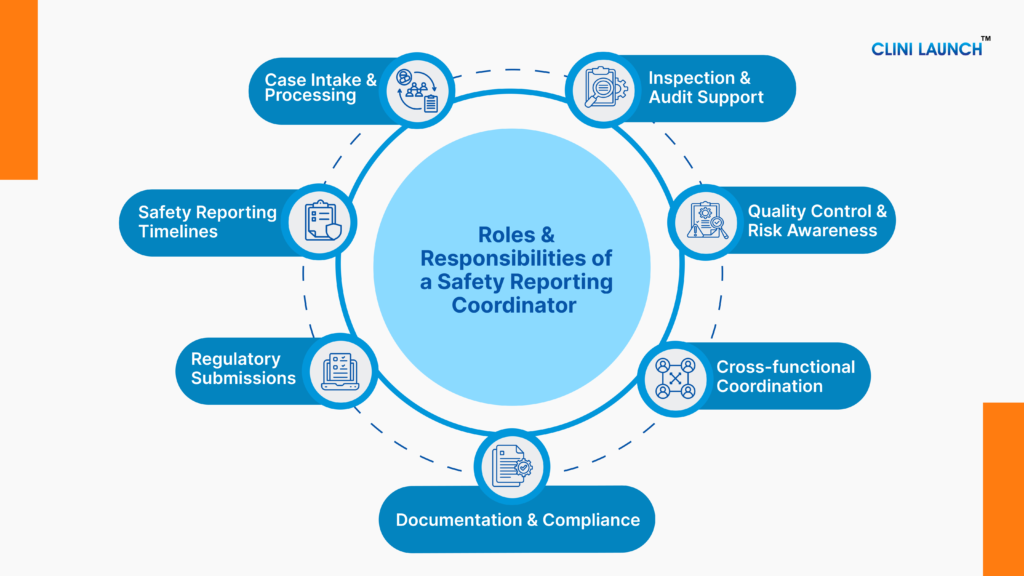
Responsibilities of a Safety Reporting Coordinator
The responsibilities of a Safety Reporting Coordinator revolve around managing safety reporting workflows, ensuring that the information flows accurately and efficiently through each required step in the process.
A. Case Intake & Processing in Clinical Safety Reporting
In this stage, the Safety Reporting Coordinator handles the initial entry of safety cases into the reporting workflow.
- Receives adverse events (AE) and serious adverse events (SAE) reports from clinical trial sites, sponsors, or CROs.
- Logs the case into the safety system using the correct study and patient identifiers.
- Performs an initial quality check to confirm that the essential details such as event description, seriousness, dates, and study of drug information are present.
- Identifies missing or unclear information and follows up with the reporting source to complete the case.
The goal of case intake and processing is to ensure that each safety case is accurate, complete, and ready for timely regulatory reporting, without delays caused by data gaps or errors.
B. Safety Reporting Timelines in Clinical Trials
Managing regulatory timelines is a core responsibility of a Safety Reporting Coordinator, particularly for GCP SAE reporting, where serious adverse events must be reported within strict regulatory timelines. Once a safety case is received and processed, it must be reported within strict, predefined timelines based on its seriousness and regulatory requirements.
- Tracks reporting deadlines for serious and expedited safety cases, including SUSARs in clinical trials.
- Monitors of case progress to ensure submissions are completed within required timeframes.
- Send reminders and follow up with internal teams or sites to avoid delays.
- Escalates potential timeline risks when required to maintain compliance.
The purpose of timeline management is to ensure that no safety report is delayed, missed or late submissions that can result in regulatory findings, audit observations, or compliance risks.
C. Regulatory Submissions in Safety Reporting
Safety Reporting Coordinators support the submission of safety reports to required stakeholders once cases are finalized.
- Coordinates the submission of safety reports to regulatory authorities, ethics committees, sponsors, and partners, as applicable.
- Ensures submissions follow approved formats and reporting pathways.
- Confirms that reports are sent within required timelines and to the correct recipients.
- Maintains submission confirmation of records for tracking and audits.
This responsibility ensures that the safety information reaches the regulatory bodies accurately on time.
D. Documentation & Compliance in Clinical Trials
Accurate documentation is essential to demonstrate compliance during audits and inspections.
- Maintains organized safety case records and supporting documentation.
- Ensure safety activities align with applicable SOPs and regulatory requirements.
- Supports internal audits, regulatory inspections, and sponsor reviews by providing required documentation.
- Tracks deviations or reporting issues and supports corrective actions when needed.
This role helps maintain a clear audit trail for all safety reporting activities.
E. Cross-functional Coordination in Clinical Research
Safety reporting involves multiple teams, and the coordinator ensures smooth communication across functions.
- Acts as a point of contact between clinical sites, pharmacovigilance teams, sponsors, and CROs.
- Coordinates information flow between data entry, medical review, and submission teams.
- Communicates case status updates and reporting requirements clearly to stakeholders.
- Supports issue resolution by facilitating timely responses across teams.
Effective coordination ensures that safety reporting processes run smoothly without delays or miscommunication.
F. Quality Control & Risk Awareness in Drug Safety
Beyond routine processing, the Safety Reporting Coordinator supports quality and risk oversight.
- Reviews cases for common data inconsistencies or reporting gaps
- Identifies potential compliance risks related to incomplete or delayed safety reporting
- Flags recurring issues for corrective or preventive action
This responsibility helps strengthen the overall quality and reliability of safety reporting.
G. Inspection & Audit Support in Clinical Trials
Safety Reporting Coordinators play an important role during audits and inspections.
- Supports regulatory inspections by providing safety case records, trackers, and documentation
- Assists in responding to audit queries related to reporting timelines and compliance
- Ensures traceability of reported safety cases during inspections
This contribution helps organizations demonstrate regulatory readiness and compliance confidence.
The table below outlines the key responsibilities of a Safety Reporting Coordinator, highlighting how safety reporting workflows are managed to ensure accuracy, compliance, and timely reporting in clinical trials.
Industry-Ready Training
| Category | Responsibility | Key Activities |
|---|---|---|
| Case Management | Case Intake & Processing in Clinical Safety Reporting | Receive AE/SAE reports, log cases, verify data, follow-up |
| Timeline Management | Safety Reporting Timelines in Clinical Trials | Track deadlines, monitor progress, send reminders, escalate risks |
| Regulatory Operations | Regulatory Submissions in Safety Reporting | Submit reports, follow formats, ensure timely delivery |
| Compliance & Documentation | Documentation & Compliance in Clinical Trials | Maintain records, follow SOPs, support audits |
| Coordination | Cross-functional Coordination in Clinical Research | Communicate with teams, coordinate workflows, resolve issues |
| Quality & Risk | Quality Control & Risk Awareness in Drug Safety | Identify errors, detect risks, ensure data quality |
| Audit & Inspection | Inspection & Audit Support in Clinical Safety | Provide records, support audits, ensure compliance readiness |
Difference between AE, SAE & SUSAR in Clinical Trials
Understanding AE vs SAE vs SUSAR is essential in safety reporting in clinical trials.
| Terms | Meanings | Examples |
|---|---|---|
| AE (Adverse Event) | Any unwanted medical occurrence | Mild headache after medication |
| SAE (Serious Adverse Event) | Life-threatening or requires hospitalization | Heart attack during trial |
| SUSAR (Suspected Unexpected Serious Adverse Reaction) | Serious + unexpected + drug-related | Unknown severe reaction not listed in protocol |
This classification helps ensure accurate and timely reporting in clinical trials. Understanding SUSAR in Clinical Research is critical, as it directly impacts regulatory reporting timelines and compliance.
Day in the life of Safety Reporting Coordinator in Clinical Trials
The day of a Safety Reporting Coordinator usually begins with checking for newly received adverse events and serious adverse event reports from clinical trial sites and partners. Each report is logged into the safety system and reviewed to confirm that essential details are present. If any information is incomplete, a follow-up request is sent immediately so the case can proceed without delay.
Once new cases are organized, attention moves to tracking regulatory timelines. The coordinator reviews which safety reports are approaching submission deadlines and monitors their progress. When risks of delay are identified, reminders are sent or concerns are escalated to ensure timelines are met.
As cases become ready, the coordinator works with pharmacovigilance teams, sponsors, and CROs to support the submission of safety reports to the appropriate authorities and ethics committees. Submission confirmations are recorded to maintain traceability and audit readiness.
Before the day ends, the coordinator ensures that safety records are up to date; outstanding follow-ups are documented, and priorities for the next day are clearly identified. Throughout the day, the role remains focused on one objective—ensuring that the safety information is accurate, timely, and properly coordinated to protect trial participants and meet regulatory expectations.
Who can become a Safety Reporting Coordinator in Clinical Trials?
A Safety Reporting Coordinator role is suitable for graduates in pharmacy, life sciences, or healthcare who are interested in safety reporting in clinical trials and pharmacovigilance.
Eligible backgrounds include:
- B.Pharm, M.Pharm, Pharm.D
- Biotechnology, Microbiology, Biochemistry, Genetics
- Nursing and Allied Health Sciences
Freshers can also enter this field with training, especially in clinical trials for safety reporting workflows. Strong attention to detail, time management, and interest in drug safety are key to success.
Salary of Safety Reporting Coordinator in India
The salary of a Safety Reporting Coordinator in India varies based on experience, organization, and skill level in clinical trials safety reporting.
- Entry-level: ₹2.5 LPA – ₹5 LPA
- Mid-level (2–5 years): ₹6 LPA – ₹10 LPA
- Experienced professionals: ₹10 LPA+
This role is commonly searched under clinical trials coordinator salary and clinical trials jobs salary India, reflecting strong interest in entry-level opportunities. Salaries are generally higher in CROs, global pharma companies, and organizations handling international trials.
Clinical Research
Build industry-ready skills to work across real clinical trial environments. Learn how clinical studies are designed, conducted, documented, and monitored, with a strong focus on ethics, patient safety, and global regulatory compliance.

Duration: 6 months
Learn at your own pace
Skills you’ll build:
Challenges faced in the role of Safety Reporting Coordinator
While the Safety Reporting Coordinator role is structured, it comes with practical challenges. Managing strict timelines is critical—for example, a serious adverse event (SAE) may need to be reported within 24 hours, leaving little room for delay.
Another common challenge is handling incomplete data. In many cases, a site may submit an SAE report without key details like event dates or lab values, requiring immediate follow-up to avoid delays in clinical trials safety reporting.
High case volumes during active study phases can increase workload pressure. Coordinators may handle multiple cases simultaneously while ensuring each report meets accuracy and compliance standards.
Additionally, global trials bring varying regulatory requirements. A report acceptable in one region may need modifications for another, requiring constant attention to submission guidelines.
Despite these challenges, structured workflows and proper training help professionals manage responsibilities effectively.
Opportunities and Career Path
The Safety Reporting Coordinator role offers a strong entry point into pharmacovigilance and clinical trials of safety reporting, with growing demand across CROs, pharma companies, and global research organizations.
Salary range:
- India: ₹2.5 LPA – ₹5 LPA (entry-level)
- With experience: ₹6 LPA – ₹12+ LPA
- Global roles: $40,000 – $80,000+ annually (depending on experience and location)
With experience, professionals can move into roles such as Senior Safety Reporting Coordinator, Drug Safety Associate, or Pharmacovigilance Officer, handling case review, submissions, and compliance oversight.
Further growth leads to roles like Safety Team Lead or Pharmacovigilance Manager, with opportunities to work on global projects. The increasing focus on drug safety compliance and regulatory reporting is driving consistent demand for skilled professionals in this field.
Career Path in Pharmacovigilance
A career in pharmacovigilance often starts with roles like Safety Reporting Coordinator and progresses into advanced positions.
Typical career path:
Safety Reporting Coordinator → Drug Safety Associate → Pharmacovigilance Officer → Safety Team Lead → Pharmacovigilance Manager
This role is a key starting point in the clinical research career path and is ideal for those exploring pharmacovigilance jobs for freshers.
With experience, professionals can also move into global safety operations, regulatory roles, or leadership positions in drug safety.
Career-Focused Guidance
Conclusion
Behind every safe and compliant clinical trial is a system of accurate and timely safety reporting in clinical trials, and the Safety Reporting Coordinator plays a key role in ensuring this process runs without errors or delays. While the role operates behind the scenes, its impact on patient safety and regulatory compliance is critical.
With increasing global focus on drug safety and regulatory reporting, the demand for skilled professionals in this field is steadily growing. If you’re looking to build a career in pharmacovigilance and clinical research, now is the right time to get started.
If you’re looking to build a strong foundation in pharmacovigilance and clinical research, CliniLaunch Research Institute offers a Advanced Diploma in Clinical Research designed to equip you with industry-relevant knowledge, practical exposure, and real-world workflows. Take the next step toward a career in clinical research to enroll today and start building your future in patient safety.
Is the Safety Reporting Coordinator role stressful?
The role involves strict timelines, but it is well-structured and manageable with good organization.
Does this role involve direct interaction with patients?
No. It focuses on safety data and reports, not direct patient interaction.
Can a Safety Reporting Coordinator work in non-clinical trial settings?
Yes. Some roles support post-marketing safety activities depending on the organization.
Is night shift or rotational work common in this role?
Some organizations require shifts, but many follow regular business hours.
How long does it take to become confident in this role?
Most professionals become comfortable within 3–6 months.
Is coding or advanced technical knowledge required?
No coding skills are required. Basic computer knowledge is sufficient.
Can this role lead to global opportunities?
Yes. Pharmacovigilance roles offer global career opportunities.
What is a Safety Coordinator’s job description?
A Safety Coordinator manages intake, tracking, and reporting of safety data.
What skills do you need to be a Safety Coordinator?
Attention to detail, time management, coordination, and communication skills.
Is the Safety Coordinator role a medical role?
No. It is a coordination and compliance-focused role.
What are pharmacovigilance jobs for freshers?
Roles include Safety Reporting Coordinator, Drug Safety Associate, and case processing roles.
What is the clinical trials coordinator salary in India?
Freshers typically earn ₹2.5–5 LPA, depending on experience.