Role of a Denial Management Specialist
Overview of Clinical Trials and Financial Accountability
Clinical trials are structured research studies designed to evaluate the safety and effectiveness of new drugs, medical devices, or treatment strategies. They follow approved study protocols, regulatory guidelines established by agencies such as the U.S. Food & Drug Administration (FDA), and defined sponsor agreements. Beyond scientific execution, clinical trials also involve detailed financial workflows. Each patient visit, laboratory test, imaging procedure, or consultation must be correctly classified as sponsor-funded research activity or routine care billable to insurance. Because trial-related services operate both research and healthcare billing systems, financial accuracy becomes as important as clinical accuracy. When billing errors occur, they do not just affect revenue — they can trigger audits, delay sponsor payments, and disrupt trial continuity. Federal agencies such as the National Institutes of Health (NIH) also outline strict guidelines for trial conduct and financial transparency.
In clinical research, claim denials are more common than many beginners expect. A significant number of trial-related claims face initial rejection due to billing mismatches, documentation gaps, or unclear responsibility between sponsors and insurers. These denials often occur even when the clinical study is conducted correctly.
For investigator sites, repeated denials lead to payment delays, increased rework, and disruption of core study activities.
Over time, this impacts site finances, delays study timelines, and strains relationships with sponsors and payers.
This challenge is expected to grow further. Industry forecasts indicate that the global healthcare denial management market is projected to grow from around USD 15 billion in 2024 to over USD 30 billion by 2030, reflecting increasing claim complexity, tighter reimbursement controls, and the rising importance of denial management in healthcare and clinical research operations.
As clinical trials become more complex and reimbursement regulations tighten, denial management has evolved into a strategic function within research operations. This shift has created demand for specialized professionals who understand both protocol compliance and revenue protection
Want to build a career as a Denial Management Specialist? Learn the complete denial management process, billing workflows, and claim analysis with practical training designed for healthcare careers.
What is Denial Management in Healthcare?
Denial management in healthcare is the structured process of identifying, analyzing, correcting, and preventing rejected or unpaid insurance claims. According to AGS Health, the denial management process involves systematically reviewing denied claims, identifying root causes, correcting errors, resubmitting claims, and implementing preventive strategies to ensure accurate reimbursements. At its core, it transforms rejected claims into paid claims while improving billing accuracy, compliance, and operational efficiency. This function is critical because unresolved denials directly impact cash flow, lead to revenue leakage, and often indicate deeper issues such as coding errors, incomplete documentation, or non-compliance with payer guidelines.
Denial management is widely used across hospitals and healthcare providers as part of revenue cycle management (RCM) and also applies to clinical research for handling trial-related billing. It focuses on managing medical claims denials, which occur due to errors like incorrect data, coding issues, missing documentation, or lack of medical necessity.
These denials can be temporary or permanent, requiring specialists to identify root causes, correct claims, and take preventive actions—making denial management a key function for improving revenue and ensuring compliance.
Denial Management Jobs & Career Opportunities in India
Denial management jobs in India are growing rapidly as healthcare organizations focus on improving revenue cycle efficiency and reducing claim losses. Demand is especially strong in major hubs like Bangalore, Hyderabad, Pune, Chennai, and Gurugram, where healthcare BPOs and RCM companies are actively hiring. This creates strong career opportunities in medical billing, AR calling, and denial analysis across healthcare operations.
- In Bangalore, there are 180+ active job openings for denial management and related roles, including AR callers and billing specialists
- Across India, platforms show 1,300+ denial management job opportunities, highlighting large-scale demand nationwide
- In Hyderabad and Pune, companies like Optum and RCM service providers actively hire professionals for denial management, appeals, and claims resolution role
Denial Management Salary Growth by Experience
| Level | Experience | Salary Range (LPA) | Typical Roles |
|---|---|---|---|
| Entry Level | 0–3 years | ₹1.8 – ₹6 LPA | AR Caller, Medical Billing Executive, Denials Specialist |
| Mid Level | 3–6 years | ₹4 – ₹8+ LPA | Denial Analyst, Senior AR Analyst, RCM Specialist |
| Senior Level | 6+ years | ₹8 – ₹15+ LPA | Team Lead, Manager – Denial Management, RCM Manager |
Entry Level
Experience: 0–3 years
Salary: ₹1.8 – ₹6 LPA
Roles: AR Caller, Medical Billing Executive, Denials Specialist
Mid Level
Experience: 3–6 years
Salary: ₹4 – ₹8+ LPA
Roles: Denial Analyst, Senior AR Analyst, RCM Specialist
Senior Level
Experience: 6+ years
Salary: ₹8 – ₹15+ LPA
Roles: Team Lead, Manager – Denial Management, RCM Manager
Hiring Sectors
- Healthcare BPO & KPO Companies
- Hospitals & Healthcare Chains
- IT & Consulting Firms
- HealthTech & AI-driven Platforms
Who is a Denial Management Specialist in Clinical Research?
A Denial Management Specialist in clinical research analyzes and resolves insurance claim denials, ensuring accurate reimbursements and compliance also known as a Denials specialist or Denial analyst.
The role focuses on managing trial-related denials by ensuring services are billed correctly to sponsors or insurance based on protocols and coverage rules. Specialists identify root causes, correct errors, and support appeals, making the role essential for maintaining compliance and protecting research revenue.
Denial Management Process in Medical Billing
The denial management process in medical billing is a structured workflow used to identify, analyze, correct, and prevent medical claims denials, ensuring accurate reimbursements and minimizing revenue loss.
Important Steps in the Denial Management Process
- Claim Submission
Submit complete and accurate claims with correct patient details, coding, and documentation.
- Denial Identification
Identify denied or rejected claims through EOB (Explanation of Benefits) or ERA reports.
- Root Cause Analysis
Determine the exact reason for denial, such as coding errors, missing information, or eligibility issues.
- Correction
Rectify errors by updating codes, patient data, or attaching necessary documents.
- Appeal
File an appeal with proper justification and supporting documentation for reconsideration.
- Prevention
Implement corrective actions and process improvements to reduce future denials.
List of Common Denial Codes in Medical Billing
Understanding common denial codes is essential in the denial management process, as it helps quickly identify the reason behind medical claims denials and take corrective action. Industry resources like Credenza Health provide detailed insights into frequently occurring denial codes and their causes.
High-Frequency Denial Codes
- CO-16 – Missing Information
This denial occurs when required details such as patient data, modifiers, or documentation are incomplete or missing.
- CO-50 – Not Medically Necessary
The payer determines that the service provided does not meet medical necessity guidelines.
- CO-97 – Included in Another Service
The service is considered part of another billed procedure and is not separately payable.
- CO-29 – Time Limit Exceeded
The claim was submitted after the allowed time frame set by the insurance provider
Clinical Research Billing Workflow and Common Denials
In clinical research, billing depends on correctly determining who pays for each service. When this classification is incorrect, medical claims denials occur and require review. Clinical trials include sponsor-paid services and routine care billed to insurance errors in this separation often lead to denials.
How the clinical research billing workflow typically works:
- Trial-related services are reviewed and classified as research-only or routine care
- Research-only services are billed to the study sponsor
- Routine care services are billed to insurance, based on coverage rules defined by payers and public insurers such as the Centers for Medicare & Medicaid Services (CMS)
- Claims are submitted along with required trial and billing documentation
- Denials arise when billing does not align with the study protocol or coverage requirements
In this workflow, denials usually fall into a few common patterns rather than random errors.
Common types of denials in clinical research billing:
- Protocol-related denials – services billed do not clearly match the approved study protocol
- Sponsor vs insurance denials – research procedures mistakenly billed to insurance
- Coverage and eligibility denials – routine care services not covered under payer policies
- Authorization-related denials – missing or incorrect approvals
- Documentation-based denials – insufficient records to support the billed service
By understanding the billing flow and common denial types, denial management specialists can quickly identify issues, take corrective action, and reduce repeated denials during clinical trials.
Since clinical research billing involves study protocols, sponsor agreements, and payer rules, denial resolution is not a routine task. It requires focused analysis and coordination across research, finance, and compliance teams making a dedicated specialist essential for accurate and compliant operations.
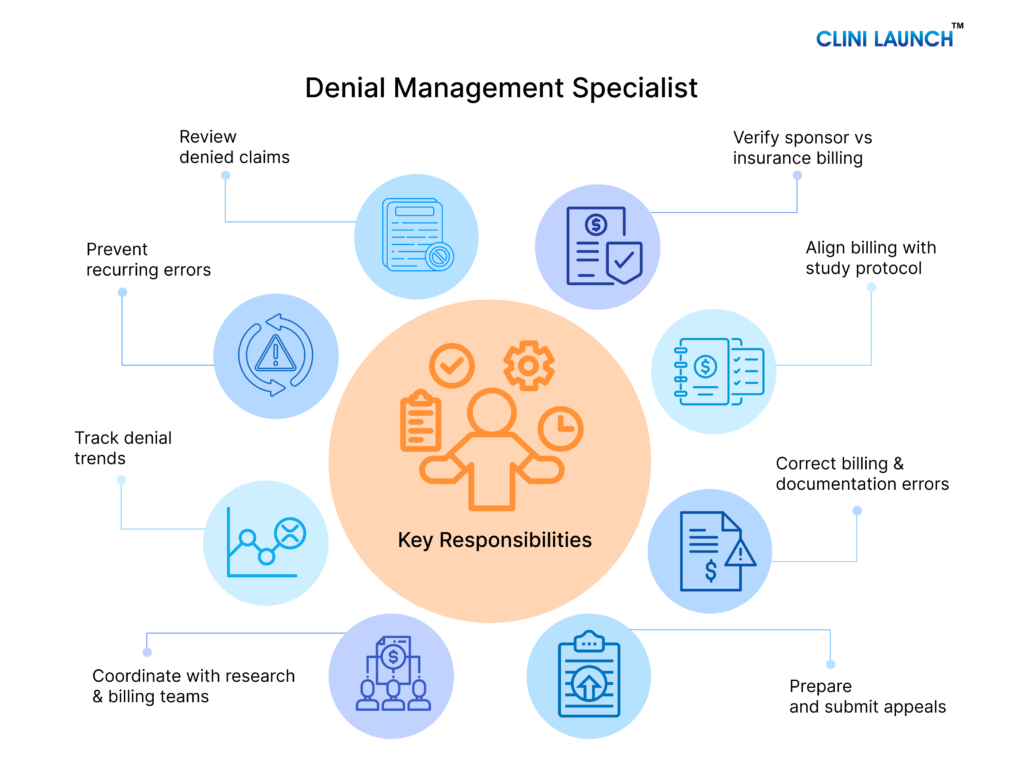
Core Responsibilities of a Clinical Research Denial Management Specialist
In clinical research, denial management is not limited to correcting rejected claims. The Denial Management Specialist plays a structured role in reviewing denials, ensuring billing accuracy, and supporting compliant reimbursement for clinical trial services. The responsibilities below outline the key areas where this role contributes to effective trial billing and financial stability for research sites and sponsors. These responsibilities collectively form the structured denial management process used to identify, analyze, resolve, and prevent billing denials during clinical trials.
- Denied Claim Review
- Sponsor vs Insurance Billing Verification
- Protocol and Coverage Mapping
- Billing and Documentation Correction
- Appeal Preparation and Submission Support
All the above responsibilities are explained briefly below.
1. Denied Claim Review
Denied claim review is the process of examining rejected claims related to clinical trial services to understand why the denial occurred and how it should be resolved. In clinical research, this step focuses on verifying whether the service was billed to the correct party, either the study sponsor or the insurance, and whether the billing aligns with the study protocol, coverage analysis, and trial agreements.
- Reconcile denied charges in the hospital billing system with the study calendar in the Clinical Trial Management System (CTMS) and the Clinical Trial Agreement (CTA)
- Review the study protocol and coverage analysis to confirm whether a service was intended to be sponsor-paid or insurance-billed
- Identify documentation or authorization gaps that contributed to the denial and coordinate with research teams to address them
This review forms the foundation for all further denial resolution and helps prevent similar billing errors from recurring during the clinical trial.
2. Sponsor vs Insurance Billing Verification
Sponsor vs insurance billing verification involves confirming that the clinical trial services are billed to the correct payer based on the study protocol, coverage analysis, and sponsor agreements. In clinical research, errors often occur when research-only procedures are mistakenly billed to insurance, or routine care services are incorrectly charged to the sponsor.
- Review study protocols, coverage analysis documents, and Clinical Trial Agreements (CTAs) to determine the correct billing pathway
- Validate whether denied services fall under sponsor-funded research activities or insurance-covered routine care
- Work with billing and research teams to correct payer assignments and re-route charges when misbilling is identified
Accurate sponsor versus insurance billing verification helps reduce unnecessary denials, ensures compliant reimbursement, and protects both investigator sites and sponsors from financial and audit-related risks.
3. Protocol and Coverage Mapping
Protocol and coverage mapping involve linking each clinical trial service to the correct billing pathway based on the approved study protocol and coverage analysis. This responsibility ensures that services performed during a trial are clearly identified as sponsor-funded research activities or insurance-billed routine care.
- Review the study protocol to understand scheduled procedures and visit requirements
- Match trial services to the coverage analysis to confirm billing responsibility
- Identify mismatches between protocol-defined services and how they were billed
Accurate protocol and coverage mapping helps prevent billing errors at the source, reduces denial risk, and supports compliant clinical research billing practices.
4. Billing and Documentation Correction
Billing and documentation correction focuses on fixing errors that lead to claim denials in clinical research billing. These errors may involve incorrect charge entries, missing or incomplete documentation, or inconsistencies between clinical records and billing information.
- Correct billing errors such as incorrect codes, modifiers, or payer assignments
- Ensure required clinical and trial-related documents are complete and accurately reflected in the billing record
- Coordinate with research and billing teams to update or resubmit corrected claims
Timely billing and documentation correction helps speed up claim resolution, supports compliant reimbursement, and reduces the likelihood of repeat denials during a clinical trial.
5. Appeal Preparation and Submission Support
Appeal preparation and submission of support involve assisting in the formal response to denied claims by providing the necessary clinical, billing, and trial-related justification. In clinical research, appeals must clearly demonstrate that the billed service aligns with the study protocol, coverage analysis, and applicable payer rules.
- Compile supporting documents such as study protocols, coverage analysis, IRB approvals, and clinical notes
- Draft or support appeal narratives explaining why the denied service is eligible for reimbursement
- Coordinate with billing teams to ensure appeals are submitted accurately and within payer timelines
Effective appeal preparation improves the chances of claim approval, reduces revenue loss, and ensures that clinical research billing decisions are properly justified and compliant.
Ready to gain real-world skills in medical billing, denial analysis, and revenue cycle management? Get hands-on training with industry tools and case-based learning.
Real-World Denial Scenarios: –
The following examples illustrate how denial management decisions directly impact clinical trial operations:
Case Study 1: Sponsor vs Insurance Billing Mismatch Leading to Claim Denial
During a clinical trial visit, a routine laboratory test was performed as part of a study-specific assessment. The claim was billed to insurance and later denied because the service was not covered under the patient’s policy.
On review, the Denial Management Specialist identified that the laboratory test was listed as sponsor-funded in the study protocol and Clinical Trial Agreement (CTA) but had been incorrectly routed to insurance. Clinical research billing policies clearly state that trial-related services must be billed to the appropriate payor based on study documentation, and misrouting charges can lead to denials
The specialist corrected the billing pathway, coordinated with the billing team to move the charge to sponsor invoicing, and aligned the documentation with the study calendar. The denial was resolved, payment was recovered, and similar billing errors were avoided for future visits.
Case Study 2: Protocol Deviation Leading to Denial of Imaging Services
During a clinical trial visit, a patient underwent an MRI scan as part of an optional exploratory assessment. The claim was submitted to insurance but was denied because the procedure did not align with the approved study protocol for reimbursable services.
The Denial Management Specialist reviewed the study protocol and coverage analysis, identified that the imaging service was optional and required sponsor pre-approval, and corrected the claim submission, ensuring future optional procedures were flagged for prior authorization to prevent repeated denials
As a result, the denied MRI claim was resubmitted, approved, and reimbursed, and the site implemented workflow checks to avoid similar denials in the future.
Tools and Documentation Used in Clinical Research Denial Management
Denial Management Specialists rely on a range of tools and documents to manage claims accurately and efficiently. Systems like CTMS, EHR, and billing software help track study procedures, patient visits, and claim status, while coverage analysis records, Clinical Trial Agreements, study protocols, and prior authorization forms provide the information needed to prevent and resolve denials. Together, these tools and documents ensure compliance, reduce errors, and support smooth billing operations in clinical research.
AI-Powered Advanced Tools in Denial Management
With increasing claim complexity and data volume, many organizations now use AI-powered revenue cycle tools to support denial management. These tools help automate claim review, detect error patterns, and predict potential denials before submission.
Many large healthcare systems and CROs now integrate denial analytics within enterprise Revenue Cycle Management (RCM) platforms. These tools use historical billing data, payer behavior trends, and protocol mapping patterns to identify high-risk claims before submission.
Examples of AI-enabled tools include:
AI-Driven Denial Analytics Platforms: Use machine learning to identify recurring denial patterns and high-risk claim categories.
Predictive Claim Scrubbing Tools: Flag billing inconsistencies before submission to reduce denial risk.
Automated Appeal Drafting Systems: Generate structured appeal templates using clinical and billing data.
Denial Prediction Dashboards: Analyze historical data to forecast denial trends across studies and payers.
These systems are increasingly integrated into enterprise RCM environments within large hospital networks and CROs, reflecting the growing role of predictive analytics in clinical billing oversight.
While AI tools improve efficiency and early detection, final review and compliance responsibility remain with the Denial Management Specialist.
Alternative Career Paths
With denial management experience, professionals can move into:
• Clinical Research Billing Specialist
• Revenue Cycle Analyst
• Compliance Officer
• Clinical Trial Finance Manager
• Healthcare Operations Manager
• RCM Technology / AI Analytics Specialist
Skills and Career Progression for Denial Management Specialist
The career progression of a Denial Management Specialist typically begins with claim review and billing verification responsibilities. With experience, professionals move into senior analytical roles, senior analytical roles, denial trend analysis of leadership and cross-functional coordination, and cross-functional coordination. Advancement depends on strengthening compliance knowledge, data interpretation skills, and workflow optimization expertise. Over time, specialists may transition into revenue cycle leadership, clinical trial finance management, or compliance-focused roles within research organizations.
Professionals aiming to build expertise in denial management and compliant billing often pursue structured training programs that combine coding, documentation, and regulatory knowledge.
Medical Coding
Build practical skills in medical coding and clinical documentation used across hospitals, healthcare providers, and global healthcare services. Learn how diagnoses, procedures, and services are accurately translated into standardized medical codes for billing, compliance, and data integrity.

Duration: 6 months
Learn at your own pace
Skills you’ll build:
Clinical Research
Build industry-ready skills to work across real clinical trial environments. Learn how clinical studies are designed, conducted, documented, and monitored, with a strong focus on ethics, patient safety, and global regulatory compliance.

Duration: 6 months
Learn at your own pace
Skills you’ll build:
Why Denial Management is critical for Clinical Research today
In modern clinical research, denial management in healthcare and clinical trials directly influences operational continuity and financial stability. When claims are denied due to protocol deviations, misbilling, missing documentation, or delayed authorizations, it can disrupt study timelines, create financial strain, and increase administrative burden for research teams. Recent data show that nearly 17 % of initial insurance claim submissions are denied, highlighting how common denials can be and why dedicated oversight is essential. A Denial Management Specialist ensures that denials are promptly identified, analyzed, and resolved, keeping trials compliant, on schedule, and financially protected. Beyond resolving individual claims, effective denial management also helps prevent recurring issues and strengthens overall billing workflows.
Key reasons:
- Protects study timelines: Resolves claim denials quickly to avoid delays in patient visits, reporting, or sponsor payments.
- Ensures financial accuracy: Minimizes lost revenue for research sites and sponsors by addressing denials efficiently.
- Maintains compliance: Ensures billing aligns with study protocols, Clinical Trial Agreements, payer rules, and regulatory standards.
- Prevents recurring errors: Analyzes denial trends to improve workflows, reduce repeated mistakes, and optimize future claim submissions.
Challenges faced in Denial Management
Despite its strategic importance, denial management presents several operational and compliance challenges:
• Complex sponsor vs insurance billing rules
• Frequent documentation gaps
• Tight appeal submission timelines
• Changing payer policies
• High denial volume & workload pressure
• Compliance and audit risk exposure
Billing misclassification in clinical trials can trigger compliance reviews under U.S. Department of Health & Human Services (HHS) oversight.
When does the role begin in a Clinical Trial?
The role of a Denial Management Specialist typically begins during the trial execution phase, when patient visits and billable services are initiated. However, involvement may start earlier during study setup, particularly in protocol and coverage mapping discussions to prevent future billing errors. The role continues through active trial conduct and remains critical during study close-out, when final reconciliations and sponsor invoicing are completed.
Take the next step toward a high-growth healthcare career. Enroll in a structured program and gain the skills needed to work in hospitals, RCM companies, and clinical research.
Conclusion
In modern clinical research, denial management directly influences operational stability and financial sustainability. By combining protocol knowledge, billing precision, and data-driven analysis, Denial Management Specialists ensure that clinical trials remain compliant, efficient, and financially secure. With the right combination of tools, documentation, and skills, these specialists not only protect research sites and sponsors but also contribute significantly to the overall success of clinical studies.
If you want to build a career in clinical research and gain the expertise needed to manage denials, claims, and trial billing efficiently, CliniLaunch Research Institute offers a comprehensive Clinical Research Course designed to equip you with practical skills and industry-ready knowledge. Join today and take the first step toward a future-ready role in clinical research.